The OptiFix™ Absorbable Fixation System has been engineered inside and out to provide surgeon confidence and secure fixation in a reliable, easy to use, and ergonomic design.


- Overview
- Products & Accessories
- EIFU & Resources

The OptiFix™ Absorbable Fastener is made from Poly(D, L-Lactide) and is designed for optimal performance. Fastener features include:
Smooth Fastener Head
• Minimises the potential for tissue attachment1
• Ensures mesh is securely fixated
Hollow Core Design
• Allows tissue ingrowth through the fastener1
Angled Tip
• Easily penetrates mesh and tissue1
Stabilizers
• Enhances tissue holding strength
• Prevents the fastener from backing out
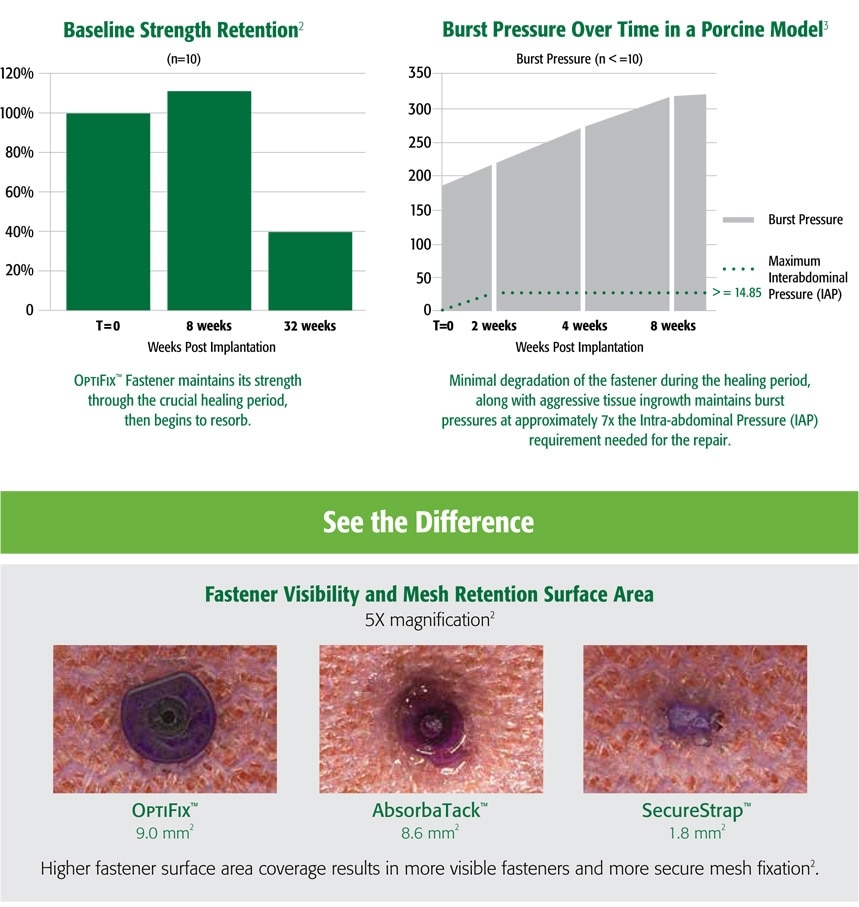
OptiFix™ Abosorbable Fasteners provide secure fixation during the postoperative healing period then slowly resorbs over time.
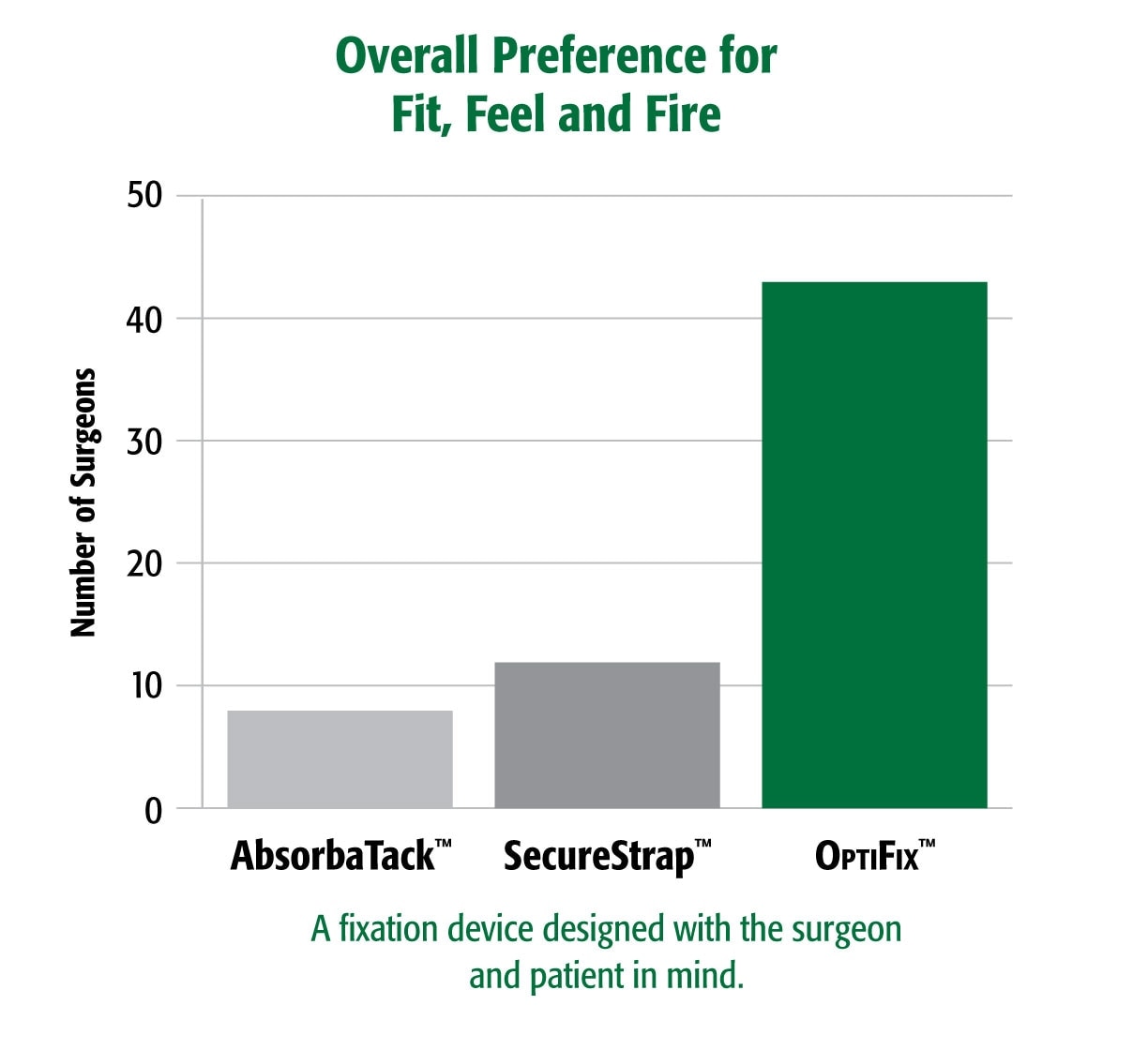
Surgeon Feedback4 Surgeons prefer the handling of OptiFix™ Absorbable Fixation System versus other fixation devices In a blind preference test assessing surgeons’ preference of device comfort, trigger force, and overall fit, feel and fire, OptiFix™ Absorbable Fixation System was preferred nearly 4x more frequently than the next closest competitor.
1. Preclinical data on file. Results may not correlate to clinical outcomes.
2. C. R. Bard Inc., bench data on file.
3. Preclinical data on file. Results may not correlate to clinical outcomes.
4. Survey of surgeons attending an international surgical conference. Data on file.
Indications
The OptiFix™ Absorbable Fixation System is indicated for the approximation of soft tissue and fixation of surgical mesh to tissues during open or laparoscopic surgical procedures, such as hernia repair.
Contraindications
1. This device is not intended for use except as indicated.
2. Do not use this device where hemostasis cannot be verified visually after application.
3. Contraindications associated with laparoscopic and open surgical procedures relative to mesh fixation
apply, including but not limited to:
• Fixation of vascular or neural structures
• Fixation of bone and cartilage
• Situations with insufficient ingrowth of tissue into the mesh over time, which could result in inadequate
fixation once the fastener is absorbed.
4. Carefully inspect the area in the vicinity of the tissue being fastened to avoid inadvertent penetration of
underlying structures such as nerves, vessels, viscera or bone. Use of the OptiFix™ Absorbable Fixation
System in the close vicinity of such underlying structures is contraindicated. For reference, the length of
the fastener below the fastener head is 6.1 mm, the fastener head is another 0.6 mm (total 6.7 mm).
5. This device should not be used in tissues that have a direct anatomic relationship to major vascular structures.
This would include the deployment of fasteners in the diaphragm in the vicinity of the pericardium,
aorta, or inferior vena cava during diaphragmatic hernia repair.
Warnings
The device may not fixate through prosthetics derived from biologic material such as xenografts and allografts. Prosthetic should be evaluated for compatibility prior to use. After use, the OptiFix™ Absorbable Fixation System may be a potential biohazard. Handle and dispose of in accordance with any local and federal laws regarding medical waste.
Adverse Reactions
Adverse reactions and potential complications associated with fixation devices such as the OptiFix™ Absorbable Fixation System may include, but are not limited to the following: hemorrhage; pain, edema and erythema at wound site; allergic reaction to Poly(D, L)- lactide; infection/septicemia; hernia recurrence/wound dehiscence.
BD-116118
1. Preclinical data on file. Results may not correlate to clinical outcomes.
2. C. R. Bard Inc., bench data on file.
3. Preclinical data on file. Results may not correlate to clinical outcomes.
4. Survey of surgeons attending an international surgical conference. Data on file.
Indications
The OptiFix™ Absorbable Fixation System is indicated for the approximation of soft tissue and fixation of surgical mesh to tissues during open or laparoscopic surgical procedures, such as hernia repair.
Contraindications
1. This device is not intended for use except as indicated.
2. Do not use this device where hemostasis cannot be verified visually after application.
3. Contraindications associated with laparoscopic and open surgical procedures relative to mesh fixation apply, including but not limited to:
- Fixation of vascular or neural structures
- Fixation of bone and cartilage
- Situations with insufficient ingrowth of tissue into the mesh over time, which could result in inadequate fixation once the fastener is absorbed.
4. Carefully inspect the area in the vicinity of the tissue being fastened to avoid inadvertent penetration of underlying structures such as nerves, vessels, viscera or bone. Use of the OptiFix™ Absorbable Fixation System in the close vicinity of such underlying structures is contraindicated. For reference, the length of the fastener below the fastener head is 6.1 mm, the fastener head is another 0.6 mm (total 6.7 mm).
5. This device should not be used in tissues that have a direct anatomic relationship to major vascular structures. This would include the deployment of fasteners in the diaphragm in the vicinity of the pericardium, aorta, or inferior vena cava during diaphragmatic hernia repair.
Warnings
The device may not fixate through prosthetics derived from biologic material such as xenografts and allografts. Prosthetic should be evaluated for compatibility prior to use. After use, the OptiFix™ Absorbable Fixation System may be a potential biohazard. Handle and dispose of in accordance with any local and federal laws regarding medical waste.
Adverse Reactions
Adverse reactions and potential complications associated with fixation devices such as the OptiFix™ Absorbable Fixation System may include, but are not limited to the following: hemorrhage; pain, edema and erythema at wound site; allergic reaction to Poly(D, L)- lactide; infection/septicemia; hernia recurrence/wound dehiscence.
1. Preclinical data on file. Results may not correlate to clinical outcomes.
2. C. R. Bard Inc., bench data on file.
3. Preclinical data on file. Results may not correlate to clinical outcomes.
4. Survey of surgeons attending an international surgical conference. Data on file.
Indications
The OptiFix™ Absorbable Fixation System is indicated for the approximation of soft tissue and fixation of surgical mesh to tissues during open or laparoscopic surgical procedures, such as hernia repair.
Contraindications
1. This device is not intended for use except as indicated.
2. Do not use this device where hemostasis cannot be verified visually after application.
3. Contraindications associated with laparoscopic and open surgical procedures relative to mesh fixation apply, including but not limited to:
- Fixation of vascular or neural structures
- Fixation of bone and cartilage
- Situations with insufficient ingrowth of tissue into the mesh over time, which could result in inadequate fixation once the fastener is absorbed.
4. Carefully inspect the area in the vicinity of the tissue being fastened to avoid inadvertent penetration of underlying structures such as nerves, vessels, viscera or bone. Use of the OptiFix™ Absorbable Fixation System in the close vicinity of such underlying structures is contraindicated. For reference, the length of the fastener below the fastener head is 6.1 mm, the fastener head is another 0.6 mm (total 6.7 mm).
5. This device should not be used in tissues that have a direct anatomic relationship to major vascular structures. This would include the deployment of fasteners in the diaphragm in the vicinity of the pericardium, aorta, or inferior vena cava during diaphragmatic hernia repair.
Warnings
The device may not fixate through prosthetics derived from biologic material such as xenografts and allografts. Prosthetic should be evaluated for compatibility prior to use. After use, the OptiFix™ Absorbable Fixation System may be a potential biohazard. Handle and dispose of in accordance with any local and federal laws regarding medical waste.
Adverse Reactions
Adverse reactions and potential complications associated with fixation devices such as the OptiFix™ Absorbable Fixation System may include, but are not limited to the following: hemorrhage; pain, edema and erythema at wound site; allergic reaction to Poly(D, L)- lactide; infection/septicemia; hernia recurrence/wound dehiscence.