Contact Support
Permanent Fixation Redefined
CapSure™ provides surgeons the confidence they desire through strong and reliable fixation.
Permanent fixation devices facilitate a strong long-term repair but may be associated with some challenges:
Covered
Strong
Reliable
*Preclinical data. Results may not correlate to the performance in humans.
316L stainless steel
Smooth PEEK cap

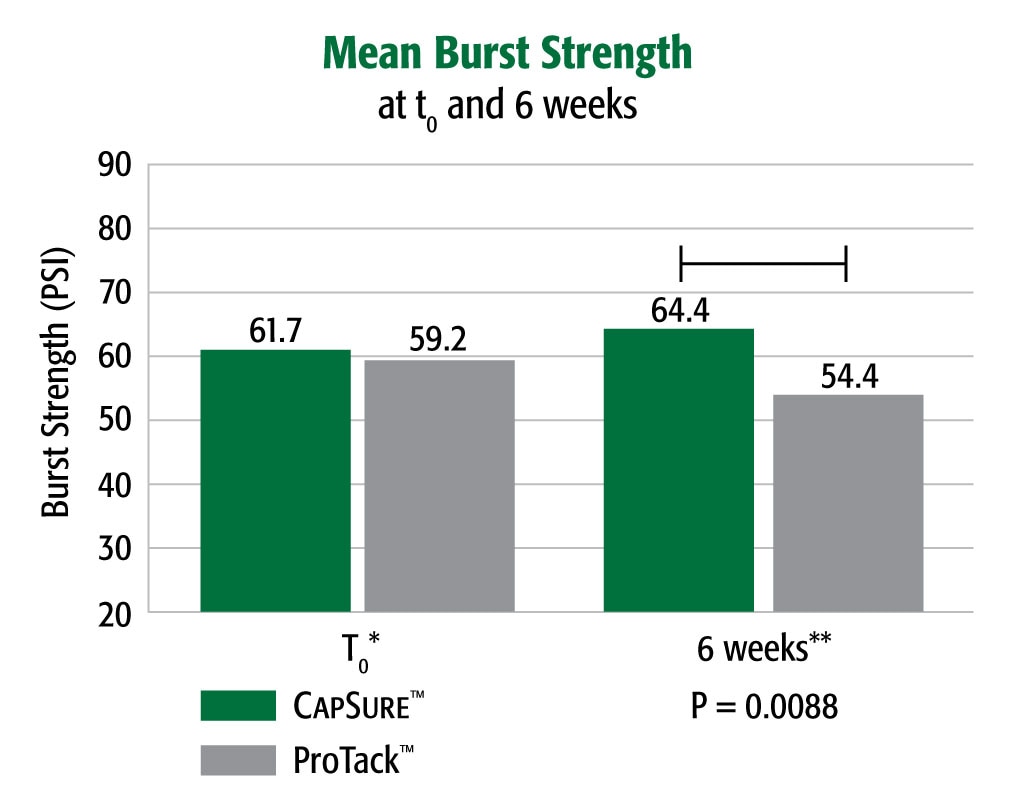
A 90 Day Preclinical Adhesion Study Demonstrated Stronger Results with the CapSure™ Fixation System vs. ProTack™ Fixation System
Evaluation of a Novel Permanent Capped Helical Coil Fastener in a Porcine Model of Laparoscopic Ventral Hernia Repair
Arnab Majumder, Mojtaba Fayezizadeh, William W. Hope,
Yuri W. Novitsky • Surgical Endoscopy April 2016
Preclinical data. Results may not correlate to performance in humans.

CapSure™ fastener easily penetrates Cooper’s ligament and underlyingstructures, equivalent to ProTack™

Burst strength testing demonstrated that in a porcine model CapSure™ fixated Ventralight™ ST Mesh had a greater (4.3%) burst strength at t0 and significantly higher (18.4%) peak burst strength at 6 weeks post-implantation than did ProTack™ fixated Ventralight™ ST Mesh at the same time point (p = .0088).
* Porcine abdominal wall tissue. Animal data may not correlate to performance in humans
* Porcine 6 week implant study. Animal data may not correlate to performance in humans
Better confidence in your delivery system performance

CapSure™ Fasteners have 2X the mesh surface area coverage to hold mesh in place ensuring secure fixation and more visible fasteners. Bench testing with 3DMax™ Light demonstrates that CapSure™ is 15X more likely to retain large pore mesh versus ProTack™1

Please note, not all products, services or features of products and services may be available in your local area. Please check with your local BD representative.
Indications.
The CapSure™ Permanent Fixation System is indicated for the approximation of soft tissue and fixation of surgical mesh to tissues during laparoscopic surgical procedures in ventral and inguinal hernia repair.
Contraindications.
Warnings.
Precautions.
Adverse Reactions.
Adverse reactions and potential complications associated with fixation devices such as the CapSure™ Permanent Fixation System may include, but are not limited to the following: hemorrhage, pain, edema and erythema at wound site; septicemia/infection; hernia recurrence/wound dehiscence, erosion and allergic response in patients with known sensitivities to PEEK and metals contained in 316L stainless steel, including chromium, nickel, copper, and iron. Please consult product package insert for more detailed safety information and instructions for use.