Contact Support
Fully Resorbable Implant for Soft Tissue Reconstruction
Phasix™ Mesh provides a fully resorbable monofilament scaffold for rapid tissue incorporation that has been designed to allow for the repair strength of a synthetic mesh along with the remodeling characteristics of a biologic graft.1
Repairs1
The open monofilament mesh scaffold provides early integration and repair strength*
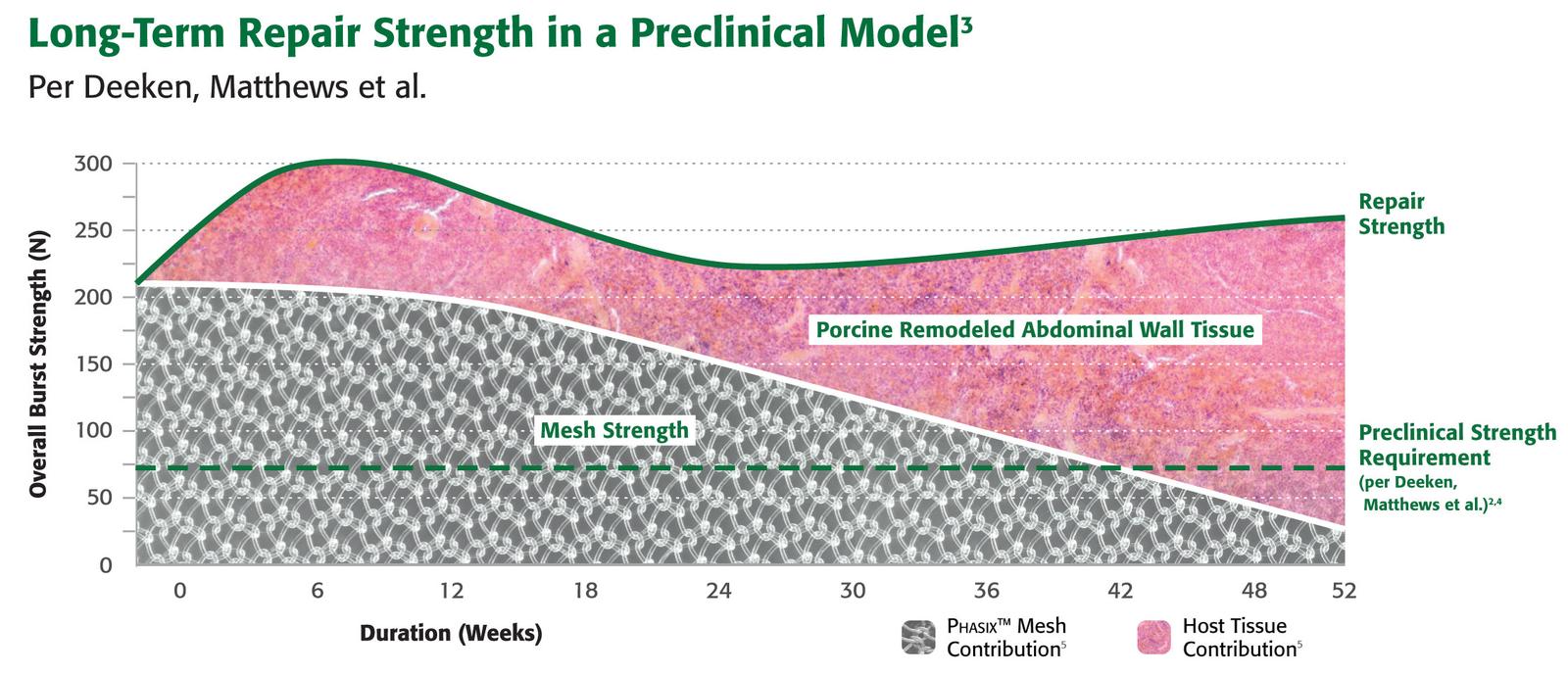
Remodels1
Preclinical testing confirms vascular integration and incorporation, with abundant mature collagen at 52 weeks. Gradually transfers load to native tissue over time*
Restores1
As Phasix™ Mesh is remodeled, it is replaced with functional tissue, ultimately resulting in a strong repair at one year*
Phasix™ Mesh is a knitted monofilament mesh scaffold using Poly-4-hydroxybutyrate (P4HB), a biologically derived, fully resorbable material.
Material structure can impact host response.5 Consider these features of monofilament versus multifilament structures:
Study objective: Evaluate material strength and histopathology of Phasix™ Mesh.
Study design: A 3-centimeter round defect was created in the ventral abdominal wall of 25 Yucatan mini-pigs (average weight 38 kg). Phasix™ Mesh was fixated directly over the defect with SorbaFix™ resorbable tacks. Ball burst testing and histopathology were conducted at 6, 12, 26, and 52 weeks.
Results: Early tissue ingrowth, vascular integration, and incorporation of Phasix™ Mesh into the ventral abdominal wall, plus abundant mature collagen formed around the remaining fibers at 52 weeks.
Prospective Multi-institutional Evaluation of Phasix™ Mesh in High Risk Ventral and Incisional Hernia Repairs:
Please note, not all products, services or features of products and services may be available in your local area. Please check with your local BD representative.
Indications.
Phasix™ Mesh is indicated to reinforce soft tissue where weakness exists, in patients undergoing abdominal, plastic, and reconstructive surgery in ventral hernia repair and other abdominal fascial defect procedures.
Contraindications.
Warnings.
Precautions.
Adverse Reactions.
In preclinical testing, Phasix™ Mesh elicited a minimal tissue reaction characteristic of foreign body response to a substance. The tissue reaction resolved as the mesh was resorbed. Possible complications may include, but are not limited to infection, seroma, pain, mesh migration, wound dehiscence, hemorrhage, adhesions, hematoma, inflammation, allergic reaction, extrusion, erosion, fistula formation and recurrence of the hernia or soft tissue defect. Please consult product package insert for more detailed safety information and instructions for use.