All of us are striving to navigate the uncertainty of the COVID-19 pandemic. As the current healthcare focus is on delivering COVID-19 vaccinations, administering rapid and accurate diagnosis and providing optimal patient care, we are leveraging our expertise and global reach to provide support and continued access to critical healthcare products. With unwavering commitment, we stand united with you—now and into the future.
Our Response Plan on COVID-19 (Coronavirus)

- Our Response Plan
- Our Responsibility in Time of Crisis
- Patient Care Support Across the Continuum
Critical Ways We Can Help You

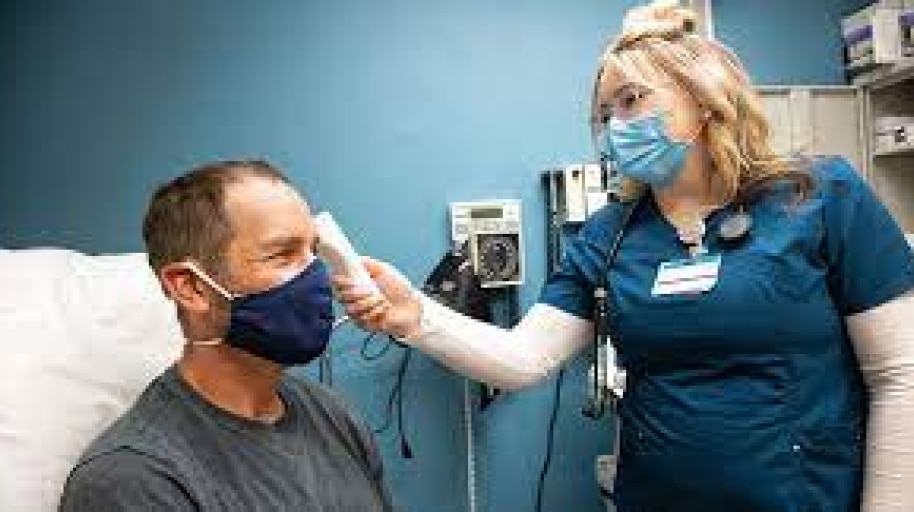
Rapid COVID-19 Testing

COVID-19 PCR Testing

COVID-19 Vaccination Preparedness
Learn More About our Efforts and Contributions
As one of the largest global medical technology companies in the world, BD is deploying its capabilities, expertise and scale to address critical health needs related to coronavirus – from diagnostic offerings to identify COVID-19, to real-time informatics and electronic surveillance technology and to essential medical devices that support patient care. This work relies on the dedication and contribution of BD employees around the world and has earned BD recognition on Fortune’s Change the World List (2020) for its fourth time in six years.
-
BD Track and Field Ad “Never Stopped”
-
65,000 people. One human hope.
-
COVID-19 Sample Collection
-
COVID-19 PCR Molecular Technology
-
COVID-19 Point of Care Antigen Testing
-
Risk of Secondary Infections in COVID-19 Patients
-
Infection Prevention and Control Training
-
Supporting COVID-19 Patients in ICUs
-
Tracking COVID-19 in the U.S
-
Supporting Research and Development of COVID-19 Treatments
-
Preparing for a COVID-19 Vaccination
-
Manufacturing
-
Government Partnerships
-
Regulatory Affairs
-
Partnering with Non-Government Organizations to Address COVID-19 Together
-
Supporting Humanitarian Organizations to Address COVID-19 Together
-
Serving the Community to Combat COVID-19
Product Training, Education and Clinical Resources
Patient management resources
COVID-19 Insights, Infection Control Training, Blog and News


Infection control training from the Society for Healthcare Epidemiology of America (SHEA) and BD

COVID-19 Blogs
COVID-19 News
Related Resources
Updated May 2022
BD continues to closely monitor COVID-19 developments across the world as well as guidance from the U.S. Centers for Disease Control and Prevention (CDC), the World Health Organization (WHO) and global health officials. We are working to protect the health and safety of BD employees while also ensuring continued availability of BD’s critical health-preserving, life-saving and diagnostic medical devices during this challenging time.
BD manufactures and sources product from multiple locations around the world. All of our global manufacturing and distribution centers are operational at this time, with the vast majority of critical-to-COVID sites operating at or near full capacity. In countries where local governments have imposed restrictions to minimize the spread of coronavirus, BD facilities continue to operate with measures in place to ensure business continuity and minimize risk of disruption to our customers.
BD continues to work closely with our key suppliers around the world that provide raw materials and components to BD manufacturing plants. We have implemented business continuity measures to mitigate the risk of potential supplier disruption, including partnering with local governments to seek “essential business” exemptions for key suppliers where necessary.
BD continues to have sufficient raw material and component inventory for the majority of products considered critical-to-COVID. Where materials or components are deemed at risk, BD activates any number of pre-approved contingency plans, including those for:
- Increased safety stock and expedited air shipments
- Seeking alternative suppliers
- Redeploying raw materials and/or finished goods from other parts of the BD network
- Placing at-risk products on customer allocation to prevent stockpiling
- Partnering with our global transportation providers and port authorities to move product through alternate routes.
Dedicated teams continue to actively monitor our global logistics and transportation network, taking action when necessary to navigate global capacity limitations and border constraints to minimize the risk of any customer delays. We are making every effort to ensure final product is transported to distributors and customers as quickly and efficiently as possible.
BD continues to follow all state, local and federal requirements and ensure adherence to work site health and safety regulations for our essential workers. We have instituted preventative measures at our facilities to help protect employees in accordance with the CDC and government regulations. We will continue to adjust safety protocols in line with these regulations and new science emerging about virus transmission and vaccine effectiveness.
As one of the largest global medical technology companies in the world, BD is deploying our capabilities, expertise and scale to address critical health needs related to coronavirus – from our diagnostic offerings to detect SARS-CoV-2, to real-time informatics and electronic surveillance technology, to essential medical devices to support patient care. Specifically, BD:
- Is collaborating with the U.S. government on the development of COVID-19 combination diagnostic tests for core labs, hospitals and point-of-care locations.
- Developed a new, rapid self-test to make COVID-19 testing faster and easier for people to complete in the privacy and safety of their own home.
- Increased manufacturing capacity in Nebraska to support U.S. vaccination agility and preparedness by providing the U.S. government with priority access to hundreds of millions of needles and syringes.
- Committed more than 2 billion needles and syringes to support global COVID-19 vaccination planning efforts in more than 40 countries and delivered shipments of more than 1 billion injection devices around the world.
- Partnered with USA Swimming to provide COVID-19 screening for the Olympic Trials and other competitive swimming events.
- Announced, along with CerTest Biotec, the VIASURE SARS-CoV-2 (N1 + N2) real time PCR detection kit for the BD MAX™ Molecular Diagnostic System that can now be run on saliva specimens.
- Partnered with USA Track & Field to provide rapid COVID-19 testing to enhance athlete safety at track & field events.
- Received Emergency Use Authorization for asymptomatic screening for SARS-CoV-2 through serial rapid antigen testing.
- Launched a rapid antigen test that can detect SARS-CoV-2, influenza A and influenza B in a single test.
In addition, BD and the BD Foundation has provided approximately $2.8 million in philanthropic support to help nearly a dozen of its trusted non-profit partners protect vulnerable communities during the COVID-19 pandemic. The majority of these social investments support healthcare workers in the United States, Europe, Latin America, China and India. BD volunteers also responded to the COVID-19 pandemic. Clinically-trained associates in BD sites around the world volunteered their time to augment hospital staff in communities hard-hit by COVID-19; and BD associates from a broad range of disciplines delivered virtual trainings to teach community and non-profit organizations how to contain its spread.

BD is a purpose-driven company, advancing the world of health™ by developing medical devices and sophisticated technologies that doctors, nurses, researchers and caregivers rely on to improve the lives of patients. As partners on the front lines, we’ve helped the world face down viral diseases for decades, from polio, smallpox and measles to HIV and the annual flu—and now, COVID-19.
Today, the capabilities, expertise and global reach of BD are greater than ever. In the face of the greatest healthcare challenge in generations, we rapidly mobilized to equip our healthcare partners with a comprehensive suite of technologies and solutions that have been essential to discovering more about the COVID-19 virus and its variants; diagnosing patients quickly, in a variety of settings; caring for those who are critically ill, and supporting the global vaccination campaign.
We continue to double-down on what we do best—tapping our deep resources to help solve healthcare’s biggest problems. We’ll never stop working with urgency to innovate, ensuring the front lines have what they need to address this crisis and prevent future outbreaks, and making an impact on lives around the world.
Tom Polen, Chairman, CEO and President
70,000 People. One Human Hope.
What We Are Doing to Address the Immediate Needs
The path forward out of COVID-19 relies on populations worldwide having ready access to tests.
BD is a critical supplier of swabs and other sample collection products for COVID-19 and has launched multiple molecular tests, enabling clinicians to know if a patient currently has COVID-19. These molecular tests are primarily completed in a hospital lab versus an offsite reference lab, so the speed to result is two to three hours, not two to three days.
Our product teams are developing new testing options that can be quickly deployed at scale, and we have already aggressively increased production of our existing portfolio to help meet the overwhelming global demand.
On any given day, our products are part of care delivery for over 90% of U.S. hospital patients.
Our technologies and devices are on the frontlines in the crisis, helping physicians and nurses manage medications, safely deliver drugs to patients and support advanced care needs in ICUs.
Our solutions, instruments, and data analysis platforms are integral to helping researchers rapidly deepen our understanding of COVID-19 at a cellular level, specifically in clarifying our picture of the body’s immune system response.
With a more detailed knowledge of immune responses, researchers have a stronger foundation to develop more effective therapies.
Carving a Path Out of the Crisis
In order to safely restart our economies, we need both multifaceted testing capabilities and advanced disease monitoring capabilities.
Our information and data technologies help health systems assess data on test results, drug inventory, hospital utilization trends and more to help form a picture of risks and responses. And as we look ahead to cautiously returning to "normal," our technologies can help detect early warning signs of re-emergence of the disease.
The race is on to mobilize vaccine administration worldwide.
BD has been by the side of vaccine developers and healthcare providers for decades, ensuring the availability and safe administration of vaccines from polio to the seasonal flu. As COVID-19 vaccinations accelerate, and researchers continue pursuing novel vaccines and treatments, BD has a full suite of medication delivery devices to meet the needs of each potential drug. We continue working with governments around the world to understand needs and scale up production.
As COVID-19 continues to challenge communities globally, BD and its associates are stepping up to respond to unprecedented needs for philanthropic and volunteer support.
BD and the BD Foundation have issued nearly $2.5 million in philanthropic grants and product donations to non-profit partners that are working to protect the most vulnerable communities and individuals from the pandemic’s spread and negative impacts. BD volunteers are heeding the call, too.
Learn about our philanthropic response:
Mobilizing to Combat this Pandemic
As one of the largest global medical technology companies in the world, BD is deploying our capabilities, expertise and scale to address critical health needs related to coronavirus—from our diagnostic offerings to identify COVID-19, to real-time informatics and electronic surveillance technology, to essential medical devices to support patient care.
Responding to crisis with action
Declared a global pandemic in March 2020, the rapid spread of COVID-19 has overwhelmed health systems and severely strained ICU capacity in hospitals. Explore how we are activating to answer the demands caused by this virus.
Expand access to COVID-19 diagnostic testing by collaborating with U.S. government agencies and molecular diagnostic companies, as well as validating alternate collection approaches.
Support patient management by facilitating complex care of critically ill patients, and providing critical devices and training to expand access to care.
Leverage informatics to track and report essential data to health agencies and providers, including near real-time insights on disease incidence, hospital utilization and capacity, and patient outcomes.
The patient is at the center of our commitment to healthcare.
-
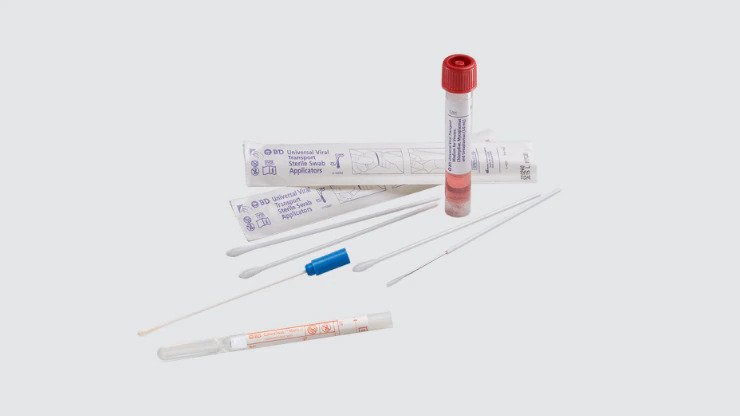
Swabs, saline, UVT medium vials and UVT kits
Used for the collection and transport of clinical specimens, including viruses.
-
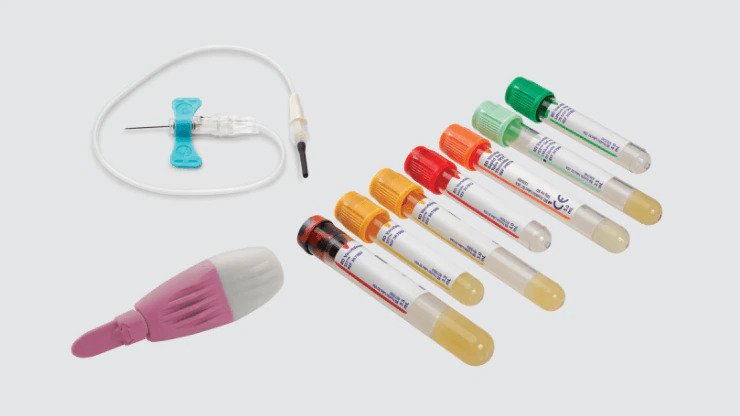
Collection sets, tubes and lancets
Used to collect blood samples for serology and blood culture tests.
-

Direct access sampling devices
Used to facilitate blood sample collection from hospitalized patients who have existing access lines.
-

BD flocked swabs
Our flocked swabs feature perpendicular nylon fibers that optimize specimen collection and elution into transport media.
-
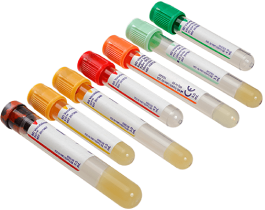
BD Vacutainer® blood collection tubes
Begin with a quality specimen for quality results
-

BD Microtainer® contact-activated lancet
The BD Microtainer® contact-activated lancet is a safety-engineered device used for the fingerstick collection of blood
-

Molecular systems
With (2) FDA EUA assays available and open system capability used to aid in the rapid detection of active COVID-19 infection.
-
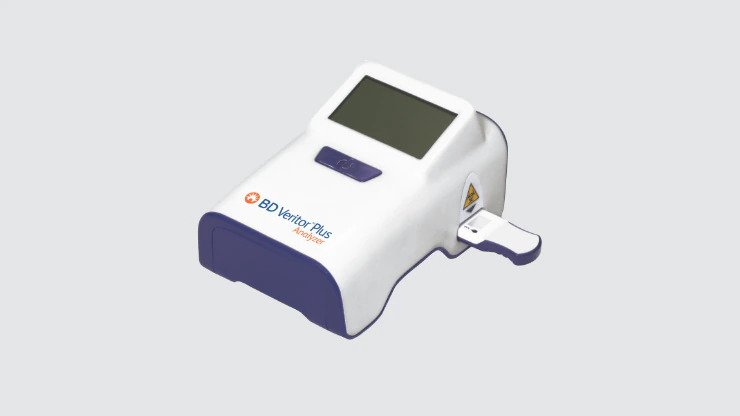
Point-of-care immunoassays
Used to rapidly detect respiratory pathogens SARS-CoV-2, Flu A, Flu B, Respiratory syncytial virus (RSV) and Group A Strep.
-

Blood culture solutions
Used to detect organisms causing bloodstream infections.
-
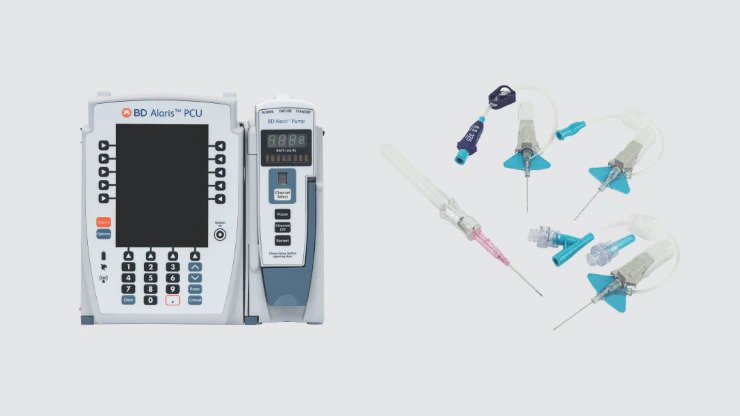
Hospital infusion pump/sets
Used for delivery of IV medications required for treatment.
-

Automated dispensing cabinets
Used to help clinicians efficiently manage medications.
-

Alaris™ Pump Module Administration Sets
Alaris™ Pump infusion sets include various configurations and a feature that integrates our SmartSite™ needle-free valve above or below the pump module.
-
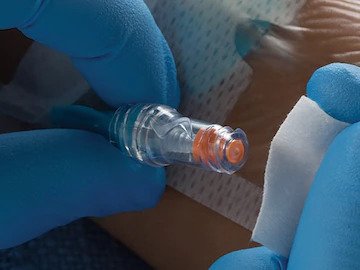
Needle-Free Connectors
Reduce the risk of IV-related infections like CLABSIs with the BD portfolio of five needle-free connectors
-
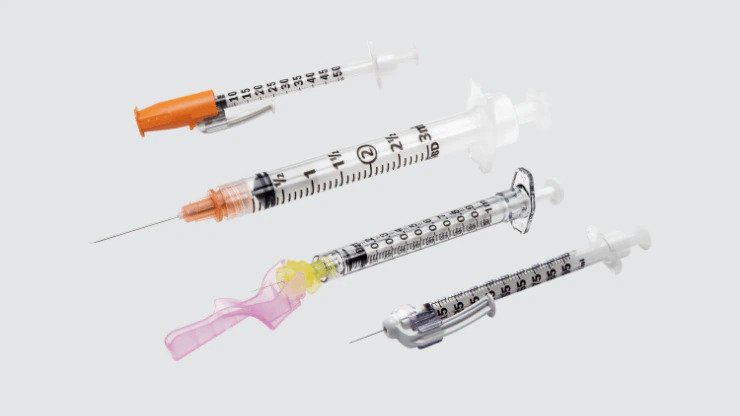
Needles and syringes
Used to administer medications required for treatment, as well as vaccination.
-
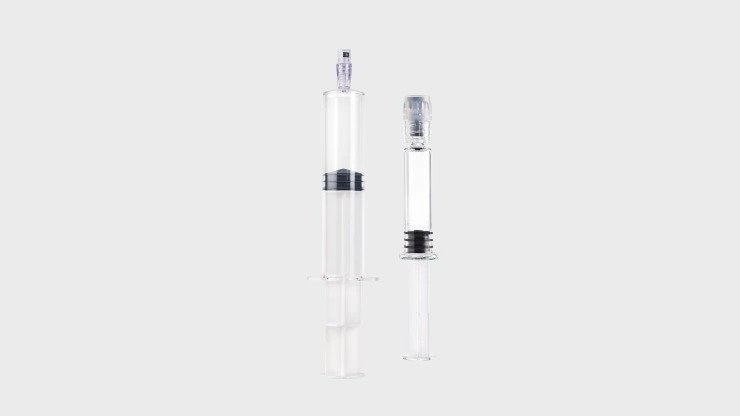
Prefillable devices
Used by the pharmaceutical industry to provide drugs and vaccines in ready-to-administer formats for speed and patient safety.
-

PowerPICC™ Provena™ Catheter
Proactive venous solutions
-
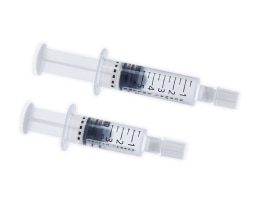
BD PosiFlush™ Pre-Filled Saline Syringe
Maintain vascular access devices
-

PICCs, midlines, and PIVCs
Used to deliver drugs required for treatment.
Care Related Products
-
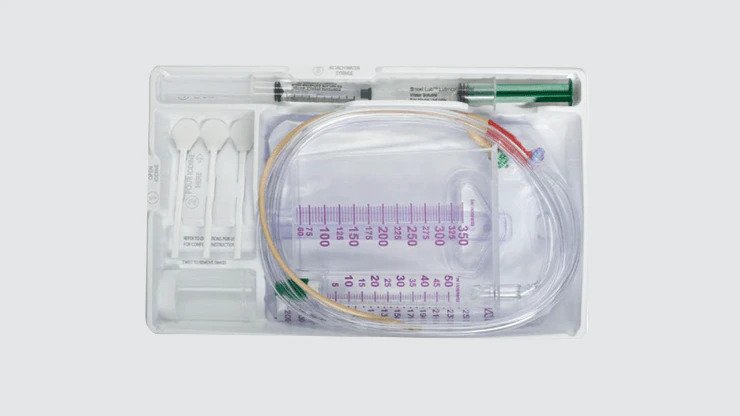
Foley catheters
Used to drain urine from the bladder in acute care settings.
-

Stool management system
Used for fecal management in bedridden patients and administration of medications.
-
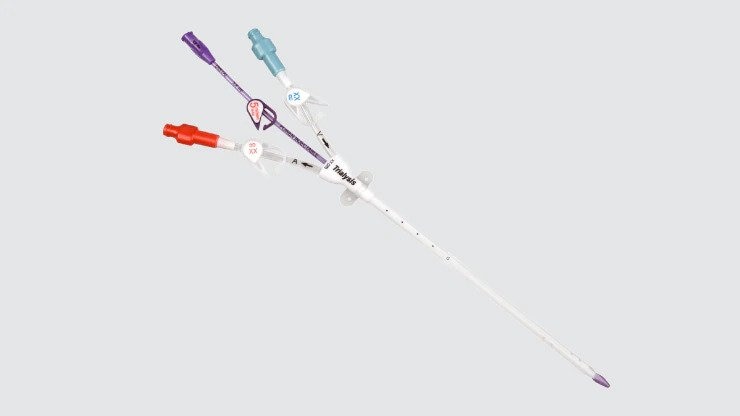
Acute dialysis catheters
Used to administer immediate dialysis for patients who enter acute renal failure.
-
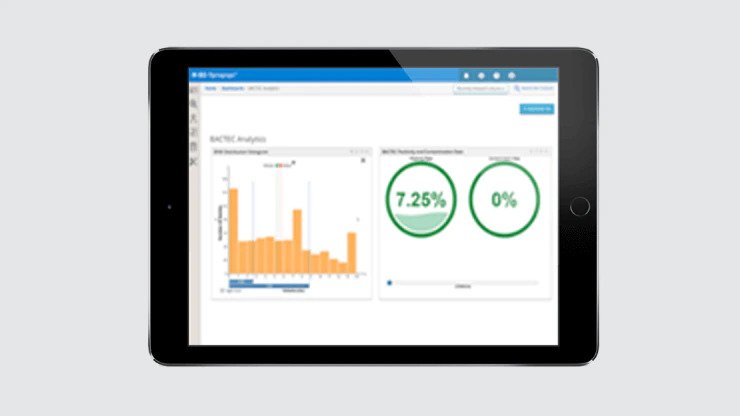
Diagnostics reporting and analytics
Used for results reporting and to provide access to actionable diagnostic information.
-
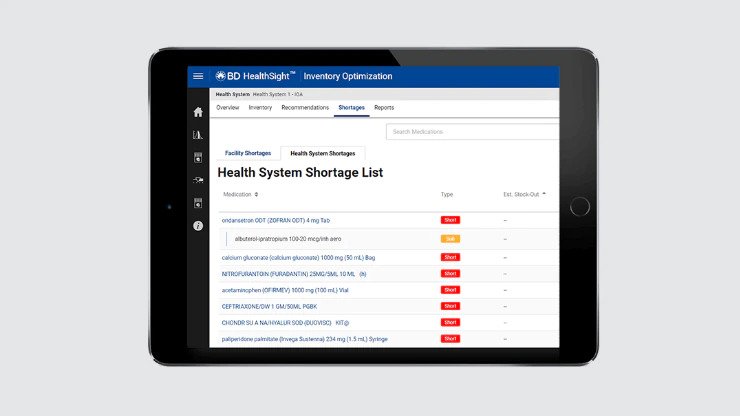
Pharmacy analytics
Used to help track and optimize medication use, including drug shortages.
-
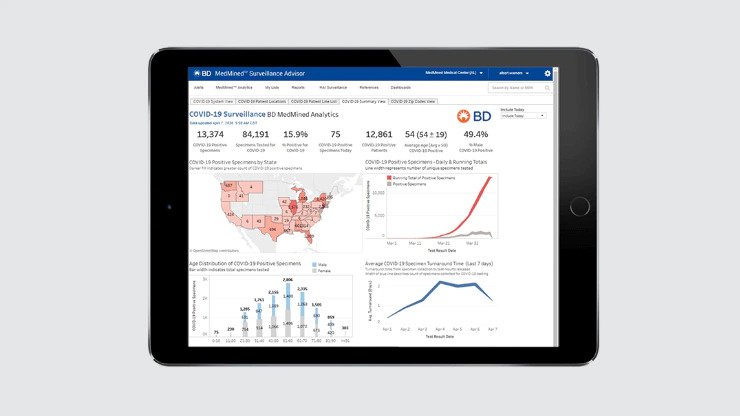
Infection surveillance analytics
Used for near real-time surveillance, reporting and understanding of geographic impact of pathogens, including COVID-19.
Not all products available in all geographies.