Contact Support
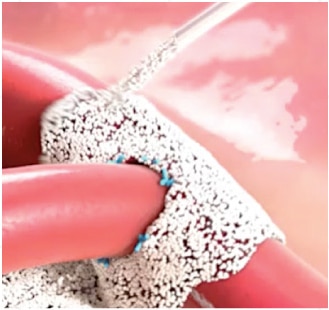
Absorbable Hemostatic Particles
Arista™ AH is a 100% plant based absorbable surgical hemostatic powder derived from purified plant starch. The power of Arista™ AH lies in its Microporous Polysaccharide Hemospheres, a patented blood clotting technology.
Arista™ AH is indicated in surgical procedures (except ophthalmic) as an adjunctive hemostatic device to assist when control of capillary, venous, and arteriolar bleeding by pressure, ligature, and other conventional procedures are ineffective or impractical.
* Because there have been reports of decreased amylase activity in newborns up to 10 months, absorption rates of Arista™ AH in this population may be longer than 48 hours.
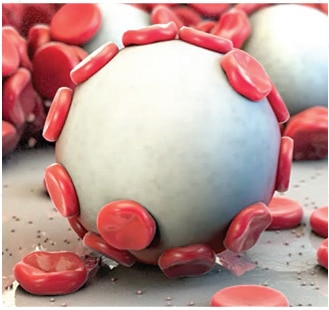
Proprietary MPH™ Technology: A Unique Approach to Achieving Hemostasis
The power of Arista™ AH lies in its proprietary MPH™ (Microporous Polysaccharide Hemospheres) technology. Consisting of microporous particles with a controlled pore size, the spheres are designed to act as a molecular sieve. The powerful osmotic action dehydrates and gels the blood on contact to accelerate the natural clotting process.

| SKU/REF | SKU/REF Name | Packaging |
|---|---|---|
| SM0002 | Arista™ AH 3 g box (absorbable hemostatic particles) | 5/cs. |
| SM0005 | Arista™ AH 1 g box (absorbable hemostatic particles) | 5/cs. |
| SM0007 | Arista™ AH 5 g box (absorbable hemostatic particles) | 5/cs. |
| SKU/REF | SKU/REF Name | Packaging (SKU) |
|---|---|---|
| AM0004 | Arista™ AH FlexiTip™ Applicator, 14 cm (includes two applicators) | 5/cs. |
| AM0005 | Arista™ AH FlexiTip™ XL Applicator, 38 cm | 5/cs. |
Please note, not all products, services or features of products and services may be available in your local area. Please check with your local BD representative.
INDICATIONS
Arista™ AH is indicated in surgical procedures (except ophthalmic) as an adjunctive hemostatic device to assist when control of capillary, venous, and arteriolar bleeding by pressure, ligature, and other conventional procedures are ineffective or impractical.
CONTRAINDICATIONS
WARNINGS
PRECAUTIONS
ADVERSE REACTIONS
In a randomized prospective, concurrently controlled clinical trial, a total of 288 randomized patients received ARISTA™ AH or the Control (Gelatin Sponge with or without Thrombin). The most common recorded adverse events were pain related to surgery, anemia, nausea, and lab values out of normal range. The following is a complete list of adverse events reported in greater than 5% of the ARISTA™ AH treated patients. The corresponding adverse events for the Control group are listed for comparison. None of the adverse events that occurred were judged by the Data Safety Monitoring Board to be related to the use of ARISTA™ AH.
Please consult product labels and inserts for any indications, contraindications, hazards, warnings, precautions, and instructions for use.
BD-51218
![]()