Same proven technique with improved mesh.
OnFlex™ Mesh was specifically designed to fit the inguinal anatomy during preperitoneal placement. It offers extended medial and inferior coverage for direct and femoral hernia spaces.
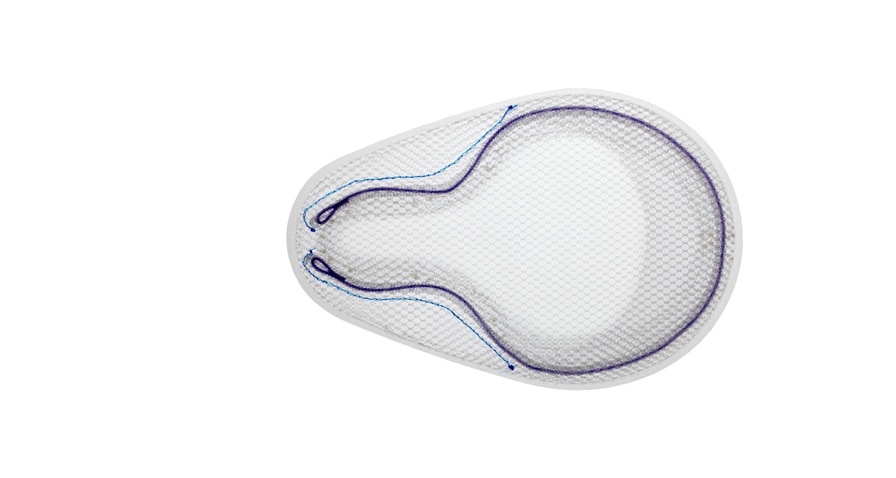

OnFlex™ Mesh was specifically designed to fit the inguinal anatomy during preperitoneal placement. It offers extended medial and inferior coverage for direct and femoral hernia spaces.
Lightweight, Large Pore Mesh
Positioning Pocket
Absorbable SorbaFlex™ Memory Technology
Interrupted PDO Monofilament
Inguinal Notch
1. Brown C, Finch J. Which mesh for hernia repair? Annals of The Royal College of Surgeons of England 2010;92(4):272-278.
* Observed in preclinical model, which may not correlate to performance in humans.
Indications
The OnFlex™ Mesh is indicated for use in the reinforcement of soft tissue where weakness exists, such as in the repair of inguinal hernias.
Contraindications
Use of this device is contraindicated for infants, children, or pregnant women, whereby future growth will be compromised by use of such mesh material. Literature reports that there is a possibility for adhesion formation when polypropylene is placed in direct contact with the bowel or viscera.
Warnings
The use of any synthetic mesh or patch in a contaminated or infected wound can lead to fistula formation and/or extrusion of the mesh and is not recommended. If an infection develops, treat the infection aggressively. Consideration should be given regarding the need to remove the mesh. Unresolved infection may require removal of the mesh.
Do not cut or reshape the Onflex™ Mesh, except at the opening in the interrupted SorbaFlex™ PDO monofilament, to accommodate the spermatic cord and outside of the blue limit line in the lateral portion of the mesh, as this could affect its effectiveness. Care should be taken not to cut or nick the SorbaFlex™ PDO monofilament.
Precautions
Care should be taken not to cut or nick the SorbaFlex™ PDO monofilament.
Adverse Reactions
Possible complications may include, but are not limited to, seroma, adhesion, hematoma, pain, infection, inflammation, extrusion, erosion, migration, fistula formation and recurrence of the hernia or soft tissue defect. If the SorbaFlex™ PDO monofilament is cut or damaged, additional complications may include, but are not limited to, bowel or skin perforation and infection.
BD's collection of literature on industry and on our offerings gives you information you can use to continue striving for excellence.
BD offers training resources to help improve your clinical practices as part of our goal of advancing the world of health.
BD supports the healthcare industry with market-leading products and services that aim to improve care while lowering costs.
BD promotes clinical excellence by providing various resources on best practices, clinical innovations and industry trends in healthcare.
Brown C, Finch J. Which mesh for hernia repair? Annals of The Royal College of Surgeons of England 2010;92(4):272-278.
Disclaimers
Not all products, services, claims or features of products may be available or valid in your local area. Please check with your local BD representative.
Please consult product labels and instructions for use for indications, contradictions, hazards, warnings, and precautions.
Indications
The OnFlex™ Mesh is indicated for use in the reinforcement of soft tissue where weakness exists, such as in the repair of inguinal hernias.
Contraindications
Do not use this mesh in infants, children, or pregnant women, whereby future growth may be compromised by the use of such mesh material.
The use of this mesh has not been studied in breastfeeding or pregnant women.
Literature reports there may be a possibility for adhesion formation when the polypropylene is placed in contact with the bowel or viscera.
Warnings
The use of any synthetic mesh or patch in a contaminated or infected wound can lead to fistula formation and/or extrusion of the mesh and is not recommended.
If an infection develops, treat the infection aggressively. Consideration should be given regarding the need to remove the mesh. An unresolved infection may require removal of the mesh.
If unused mesh has been in contact with instruments or supplies used on a patient or contaminated with body fluids, discard mesh with care to prevent risk of transmission of viral infections.
To avoid injury, careful attention is required if fixating the mesh in the presence of nerves, vessels or the spermatic cord. Fastener penetration into underlying tissue containing nerves or blood vessels may result in the need for medical/surgical intervention, cause serious injury or permanent impairment to a body structure.
This mesh has been designed for single use only. Reuse, reprocessing, resterilization or repackaging may compromise the structural integrity and/or essential material and design characteristics that are critical to the overall performance of the mesh and may lead to device failure which may result in injury to the patient. Reuse, reprocessing, resterilization or repackaging may also create a risk of contamination of the mesh and/or cause patient infection or cross infection, including, but not limited to, the transmission of infectious diseases from one patient to another. Contamination of the mesh may lead to injury, illness or death of the patient or end user.
This device is not for the use of repair of pelvic organ prolapse.
This device is not for the use of treatment of stress urinary incontinence.
The safety and effectiveness of OnFlex™ Mesh in laparoscopic inguinal hernia repair procedures has not been evaluated or established.
To prevent recurrences when repairing hernias, the mesh should be sized with appropriate overlap for the size and location of the defect, taking into consideration any additional clinical factors applicable to the patient. Careful attention to mesh fixation placement and spacing will help prevent excessive tension or gap formation between the mesh and fascial tissue.
The mesh is supplied sterile. Inspect the packaging to be sure it is intact and undamaged prior to use.
Do not cut or reshape the OnFlex™ Mesh, except at the opening in the interrupted SorbaFlex™ PDO monofilament, to accommodate the spermatic cord and outside the blue limit line in the lateral portion of the mesh, as this could affect its effectiveness. Care should be taken not to cut or nick the SorbaFlex™ PDO monofilament.
Precautions
Please read all instructions prior to use.
Only physicians qualified in the appropriate surgical techniques should use this mesh.
If fixation is used, care should be taken to ensure that the mesh is adequately fixated. If necessary, additional fasteners and/or sutures should be used.
Care should be taken not to cut or nick the SorbaFlex™ PDO monofilament.
Adverse Reactions
Possible complications may include, but are not limited to, seroma, adhesion, hematoma, pain, infection, inflammation, extrusion, erosion, migration, fistula formation, allergic reaction, and recurrence of the hernia or soft tissue defect. The mesh must be removed immediately if the SorbaFlex™ PDO monofilament is cut or damaged during insertion of fixation. If the SorbaFlex™ PDO monofilament is cut or damaged, additional complications may include, but are not limited to, bowel or skin perforation and infection.
BD-60732 (09/22)