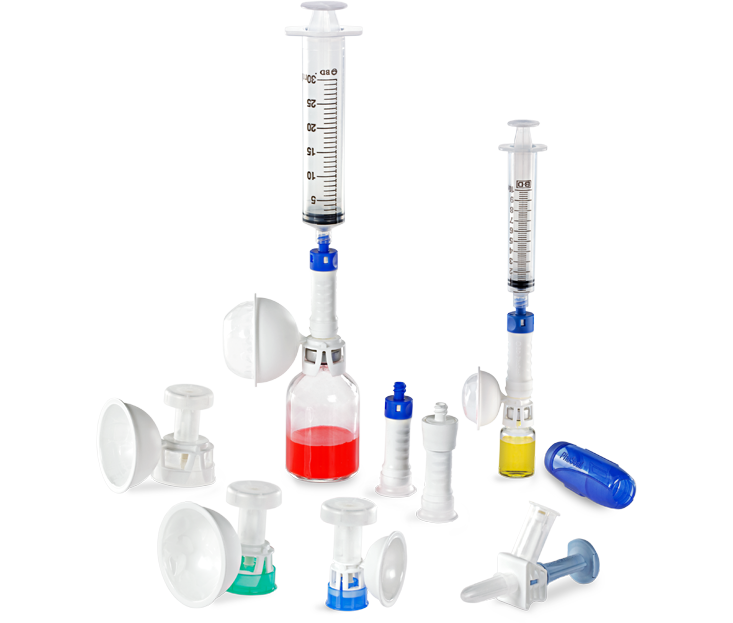
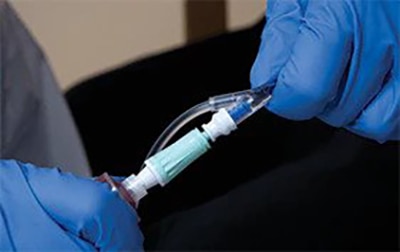
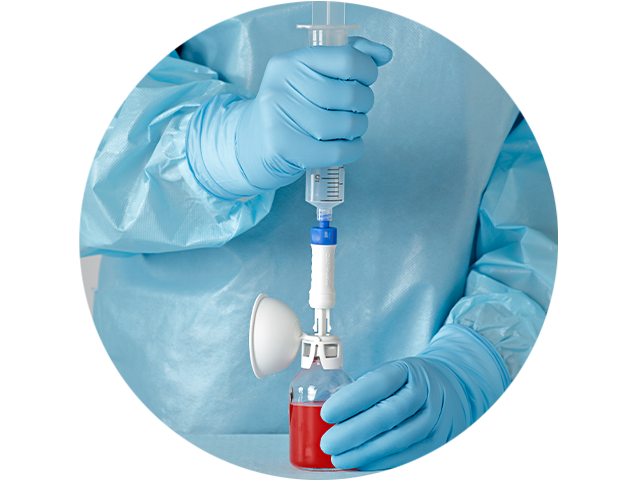
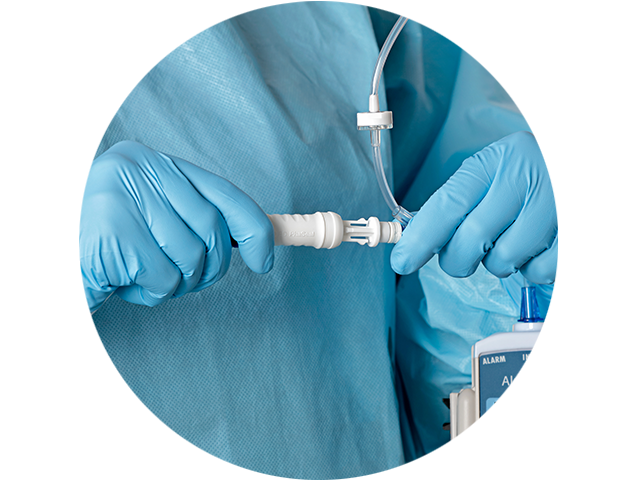
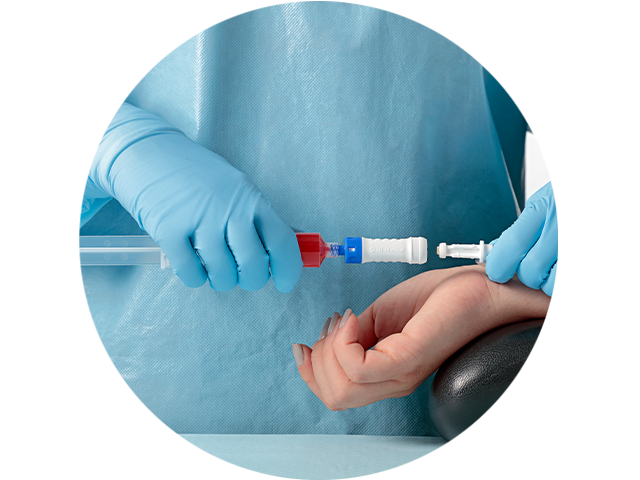
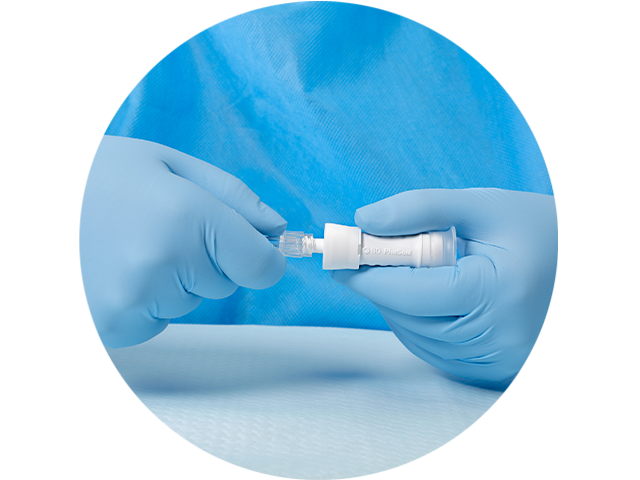
The PhaSeal™ Optima System, a second generation Closed System Drug Transfer Device (CSTD), retains the architecture of the PhaSeal™ System and utilises the same physical barrier technology to help prevent hazardous drug exposure.1 This includes a vapour-capturing mechanism integrated into the vial protector and self-sealing, membrane-to-membrane connections that maintain an airtight, leakproof seal—even upon disconnection—ensuring safe and secure drug transfer.2-4
The PhaSeal™ System is the first Food and Drug Administration (FDA) cleared CSTD under the ONB product code for both preparation and administration.5,6