Should healthcare workers be concerned about exposure to hazardous drugs?
Healthcare workers – pharmacists, technicians, nurses – work every day to improve patients’ health. However, exposure to the same hazardous drugs they prepare, handle and administer to patients may put their own health at risk.¹
Despite well-established safety standards and guidelines from organisations such as the United States Pharmacopeia (USP),2 Institute for Occupational Safety and Health (NIOSH)1 and the Occupational Safety and Health Administration (OSHA),3 hazardous drug contamination remains a persistent challenge. It continues to occur frequently and can easily spread throughout healthcare institution.4
How can you help prevent exposure to hazardous drugs?
The recent EU Directive 2022/4315 on protecting workers from exposure to carcinogens or mutagens in the workplace requires the implementation of appropriate procedures to monitor these substances. This includes ensuring early detection of abnormal exposure – particularly in the event of unforeseen incidents or accidents – through established measurement protocols.
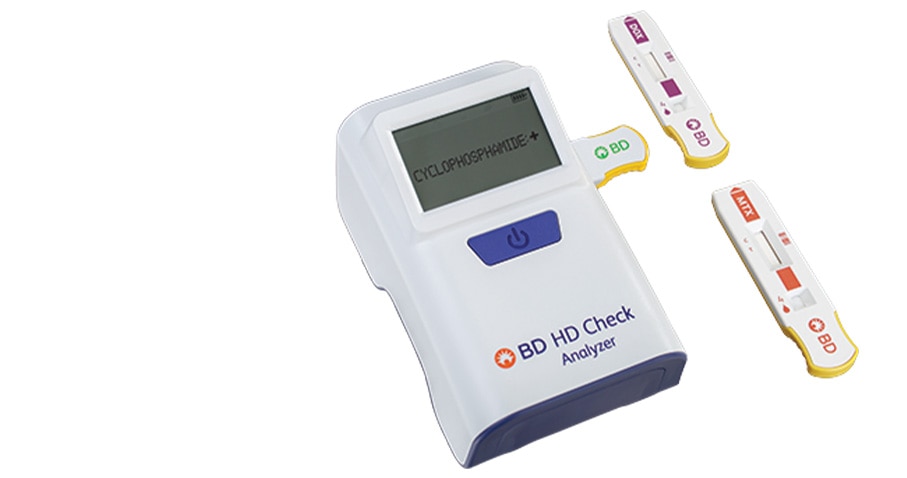
What’s the BD® HD Check System?
The BD® HD Check System is a breakthrough in hazardous drug detection*, offering the first rapid testing solution that delivers results in under 10 minutes.6,7 Designed for routine monitoring, it enables immediate corrective action when contamination is detected.8 Easy to use and fast-acting, the BD® HD Check System helps healthcare institutions enhance safety with confidence.9,10
*The BD® HD Check System tests for select hazardous drugs - cyclophosphamide, doxorubicin and methotrexate. Surfaces with contamination at or above the limits of detection have 95% specificity and sensitivity.