

- Overview
- Product Details
- Assays
A New Dawn in Molecular Diagnostics

The BD COR™ System is the first truly integrated high-throughput molecular diagnostic platform designed to transform laboratory operations. By combining pre-analytical and analytical automation, the BD COR™ System is designed to help labs overcome staffing shortages, economic challenges, and quality control needs—while delivering accurate, timely results.
Breaking News!
Onclarity™ Self-collection* Kit for At-Home Use is NOW FDA-cleared!
*Self-collected vaginal specimens can be tested as an alternative specimen type when cervical sampling is either contraindicated or cervical samples otherwise cannot be obtained.
Modern labs face increasing demands.
The fully automated BD COR™ System addresses these challenges by:
-
Reducing staff burden by automating more steps from pre-analytics to reporting
-
Delivering accurate clinical answers with high-demand women’s health and infectious disease tests
-
Providing expert support from dedicated support teams
Integrated, Scalable Automation:
BD COR™ System offers modular flexibility to meet your lab’s needs

Improve lab efficiency and clinical outcomes.

Details of the BD COR™ System
- Fully automated pre-analytics with onboard rehydration.
- Modular configurations for scalable growth.
- Expanded access to high-performance tests for women’s health and infectious diseases.
Click here to see all product details.
A hands-off approach to high-volume testing
The future is fully automated.
Save time and resources with end-to-end integrated automation.
Free your staff to focus on more complex work by automating these pre-analytical tasks:
- Sample sorting
- Vortexing
- Uncapping
- Onboard rehydration
- Aliquoting
- Recapping
- Pre-warming or cooling
- Tube and rack handling
Benefits for all:
- Labs: Streamlined operations by processing more samples with fewer steps and less staff effort.1-4
- Clinicians: Faster, more reliable results to guide timely treatment.2-5
- Patients: Quick, accurate diagnoses designed to improve better outcomes.2-7
Capacity and Throughput
Scale your BD COR™ system to fit your needs.
Configuration Option | BD COR™ Single GX System
| BD COR™ Single MX System | BD COR™ Dual GX System | BD COR™ Dual MX System | BD COR™ GX/PX/MX System |
|---|---|---|---|---|---|
Throughput | Mid to high volume for HPV | Mid to high volume throughput for vaginitis, prevalent STIs and GI testing | High throughput volume for HPV | High throughput volume for vaginitis, prevalent STIs and GI testing | Labs performing wide range of multiplex assays |
Results per 24 hours | 540 | 1,000* | 1,080 | 2,000* | 540 HPV results 1,000* molecular results |
Results in one 8-hour shift | 3305 | 6484* | 6605 | 1,2484* | 330 HPV results 648 molecular results2,3* |
*BD COR™ MX System throughput numbers are for the BD CTGCTV2 assay and may vary with other assays and specimen types.
*BD COR™ MX System throughput numbers are for the BD CTGCTV2 assay and may vary with other assays and specimen types.
See the full assay menu for the BD COR™ System

Smaller Footprint. Bigger Impact.
The BD COR™ System has a 48- 58% smaller footprint than other systems that require tracks to integrate pre-analytical automation.8-11 The BD COR™ System fits flush to the wall, giving you freedom to optimize your lab layout.
BD COR™ System Assays
Capacity that keeps up with care. Ensure clinicians have uninterrupted access to testing and patients receive results without delay—even when demand surges.
Explore the assay portfolio:

NEW! FDA Cleared Onclarity™ Self‑Collection Kit for At-Home Use
Introducing an at‑home self‑collection* kit designed to meet patients where they are while keeping clinicians anchored in innovation — with the BD Onclarity™ HPV testing with extended genotyping for risk-based patient management.
*Self-collected vaginal specimens can be tested as an alternative specimen type when cervical sampling is either contraindicated or cervical samples otherwise cannot be obtained.
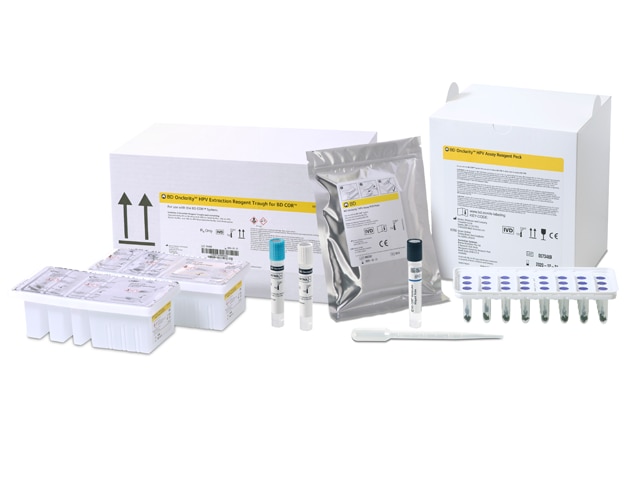
BD Onclarity™ HPV Assay
BD Onclarity™ HPV Assay is the only FDA-approved test with both a self-collection* option and extended genotyping beyond HPV 16, 18, 45.
It supports advanced risk stratification to detect persistent HPV infections before they turn cancerous and reduces barriers to screening with self-collection* so any woman, anywhere†, can test.
Let’s redefine cervical cancer screening. Discover how you can offer this to your providers.
*Self-collected vaginal specimens can be tested as an alternative specimen type when cervical sampling is either contraindicated or cervical samples otherwise cannot be obtained.
†When under the supervision of a licensed healthcare provider.
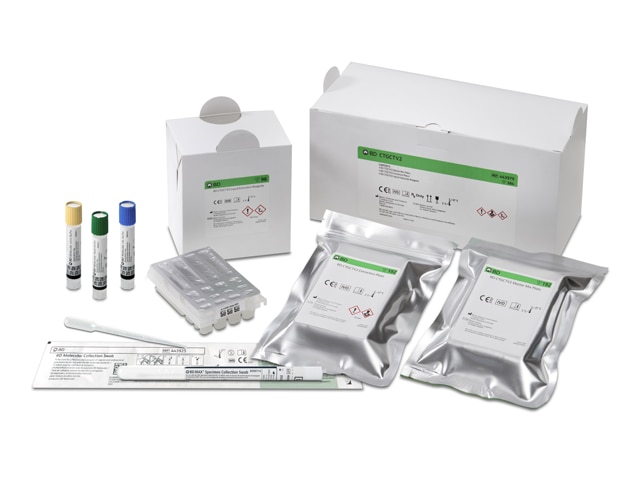
BD CTGCTV2 for BD COR™ System
The BD CTGCTV2 assay simultaneously detects the three most common non-viral STIs, Chlamydia, Gonorrhea, and Trichomonas, through a single sample. This assay goes one step further than many other STI panels by including TV in the same assay, which has a higher prevalence than Chlamydia and Gonorrhea combined. BD CTGCTV2 adapts to patient needs with a wide variety of specimen collection types available - Vaginal Swab (self-collected in a clinical setting or clinician collected), Urine, Endocervical*, Oropharyngeal*, Rectal*, and PreservCyt(R) LBC*. Explore the possibilities of testing trichomoniasis at the same time as GT/GC on the BD CTGCTV2 assay.
*CT and GC only
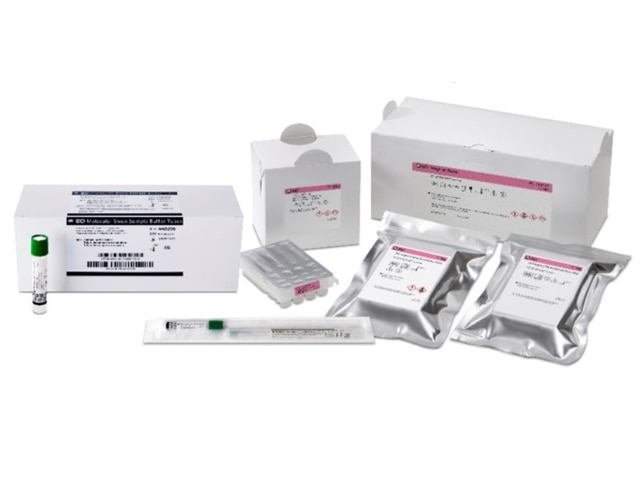
BD Vaginal Panel for
BD COR™ System
The BD Vaginal Panel is a comprehensive diagnostic test that directly detects the three most common infectious causes of vaginitis – bacterial vaginosis (BV), vulvovaginal candidiasis (VVC), and Trichomonas vaginalis (TV) – in one test, using a single swab. This panel is specifically designed to help eliminate ambiguity in test results by providing clear positive or negative outcomes for each condition. Additionally, it delivers separate outcomes for Candida glabrata and C. krusei, two Candida species known for their resistance to conventional antibiotics. Find out more about how BD Vaginal Panel assay is shaping the future of Women's Health.
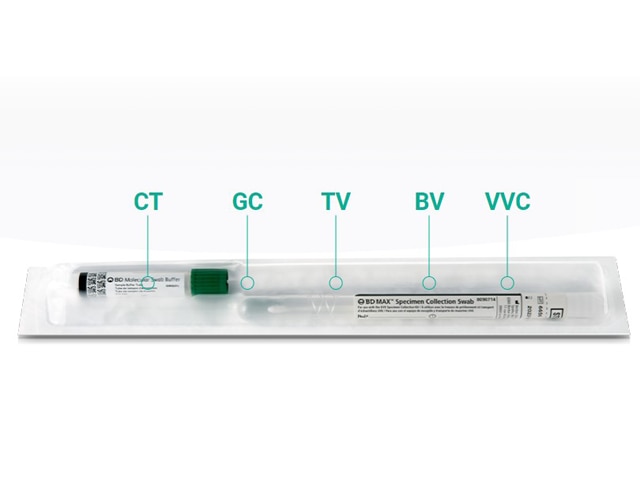
1 Sample, 7 Results
BD Vaginal Panel and BD CT/GC/TV2 for BD COR™ System can use the same sample to test for vaginitis and the 3 most common non-viral STIs in symptomatic women. 1 Sample, 5 Conditions, 7 Results. Explore more about how BD Vaginal Panel and BD CTGCTV2 can be used together to help shape the future of Women's Health.
BD EBP for BD COR™ System
The BD Enteric Bacterial Panel for BD COR™ System enables automated, direct qualitative detection and differentiation of key enteric bacterial pathogens, including Salmonella spp., Campylobacter spp. (C. jejuni and C. coli), Shigella spp./Enteroinvasive Escherichia coli (EIEC), and Shiga toxin genes (stx1/stx2) associated with Shiga toxin–producing E. coli (STEC). Testing is performed using real-time PCR technology on the BD COR™ System, providing high sensitivity and specificity for symptomatic patients suspected of acute gastroenteritis, enteritis, or colitis. The assay is seamlessly integrated with BD Fecal Collection and Transport devices for unpreserved soft to diarrheal stool specimens. Talk to a sales rep to learn more.
BD EBP plus for BD COR™ System
The BD Enteric Bacterial Panel plus for BD COR™ System expands detection capabilities beyond the standard panel, to additional lower prevalence, clinically relevant enteric pathogen for comprehensive gastrointestinal testing. This panel has the same targets as the standard panel and also includes Plesiomonas shigelloides, Vibrio spp. (V. vulnificus, V. parahaemolyticus, and V. cholerae), Enterotoxigenic Escherichia coli (ETEC) heat-labile enterotoxin (LT) / heat-stable enterotoxin (ST) genes, and Yersinia enterocolitica. The test is performed on the BD COR™ System and is seamlessly integrated with BD Fecal Collection and Transport devices for unpreserved soft to diarrheal stool specimens.
BD Diagnostic Solutions is Now Waters Advanced Diagnostics
BD Diagnostic Solutions and Biosciences have combined with Waters Corporation to create a life sciences and diagnostics leader driven by innovation and a relentless commitment to our customers.
As we begin our new journey as part of Waters, there will be no immediate changes in how we serve you. The way you order your products and services and how you process payment for them will not change. Your points of contact for sales, customer service, technical service and support or clinical services will remain the same.
References:
1. Ejegod DM, Pedersen H, Pedersen BT, et al. Clinical Validation of the Onclarity Assay After Assay Migration to the High-Throughput COR Instrument Using SurePath Screening Samples From the Danish Cervical Cancer Screening Program. Am J Clin Pathol. 2022;157(3):390-398. doi:10.1093/ajcp/aqab138
2. HTMI-22-0004 - MX Throughput Capping
3. Taylor SN, Eckert K, Rucki AA, et al. Evaluation of the Onclarity HPV assay on the high-throughput COR system.Expert Rev Mol Diagn. 2021;21(3):333-342. doi:10.1080/14737159.2021.1894132
4. HTDI-21-0406 BD COR™ Hands-On Setup Time
5. Vaughan LM and Malinowski DP. Rev Bras Ginecol Obstet. 2019;41(5):357–9.
6.Gargano JW, Meites E, Watson M, Unger ER, Markowitz LE. Human papillomavirus. In: Manual for the Surveillance of Vaccine-Preventable Diseases. 5th ed. Centers for Disease Control and Prevention; 2022. Accessed March 23, 2025. https://www.cdc.gov/surv-manual/php/table-of-contents/chapter-5-human-papillomavirus.html
7. Fuzzell LN, Perkins RB, Christy SM, Lake PW, Vadaparampil ST. Cervical cancer screening in the United States: Challenges and potential solutions for underscreened groups. Prev Med. 2021;144:106400. doi:10.1016/j.ypmed.2020.106400
8. REF-36167 COR PXGX System User Manual.pdf
9. Roche. cobas® 6800 System. https://diagnostics.roche.com/us/en/products/instruments/cobas-6800-ins-2693.html.Accessed May 14, 2025.
10. Roche. cobas® 8800 System. https://diagnostics.roche.com/us/en/products/instruments/cobas-8800-ins-2694.html.Accessed May 14, 2025.
11. Roche. cobas® Prime pre-analytical system specifications. https://diagnostics.roche.com/content/dam/diagnostics/Blueprint/en/pdf/rmd/roche-cobas-prime-pre-analytical-system-specifications-sheet_EN.pdf. Accessed May 21, 2025
Disclaimer:
This business has been acquired by Waters Corporation (“Waters”). For products referenced on this page, the legal manufacturer remains Becton, Dickinson and Company or one of its affiliates or subsidiaries (“BD”) until all required regulatory transfers are completed. During this interim period, BD maintains full responsibility for all regulatory obligations of the legal manufacturer. Product information provided here is supplied under the regulatory authority of BD. To learn more about the relationship between Waters and BD during this transition period, please see our detailed summary waters.com/bdtransaction.