-
HPV CLICK CARD 1000
SKU/REF 443747
-
BD Viper™ LT System Instrument Manual
SKU/REF 443506
-
BD Viper™ LT Waste Bag
SKU/REF 442968
-
BD Viper™ LT SDA Accessory Kit
SKU/REF 442958
-
BD Viper™ LT PCR Tube/Carrier Accessory Kit
SKU/REF 442957
-
BD Viper™ LT System
SKU/REF 442839
-
BD Viper™ SDA Extraction Reagent Trough
SKU/REF 441994
-
BD Viper™ LT Seismic Table
SKU/REF 441983
-
Swab Diluent for the BD ProbeTec™ Qx Amplified DNA Assays
SKU/REF 441361
-
BD Viper™ 2 Gallon Liquid Waste Bottle
SKU/REF 441351
-
BD Viper™ One Gallon Liquid Waste Bottle
SKU/REF 441072
-
BD Viper™ Microwell Package 8 EA
SKU/REF 440752
BD Viper™ LT system
Be her advocate for more precise HPV testing with extended genotyping to identify HPV types beyond 16, 18 and 45


- Overview
- Products & Accessories
- EIFU & Resources
Automated molecular testing
BD Onclarity™ HPV assay testing in a compact, integrated self-contained table-top unit
The BD Viper™ LT system offers fully automated molecular testing for the BD Onclarity™ HPV assay – the only FDA-approved assay with extended genotyping – out of BD SurePath™ Liquid-based Pap Test and the Hologic ThinPrep® Pap Test.
C30 sample results in
4.5 hours1
<20 minutes of total
hands-on time per run1

associated products
BD Onclarity™ HPV Assay
- Extended genotyping
Can individually identify HPV 31, which poses a higher risk for cervical precancer as compared to HPV 18
- Risk factor traceability
Can track genotype-specific high-risk HPV persistence, the most important determinant of cervical cancer risk in women who test HPV-positive, regardless of HPV genotype.
Efficiency
- Compact, integrated, self-contained table top system
- Room temperature reagent storage
- Sample to results within a single system
Flexibility
- Supports bi-directional LIS integration
- Middleware solution through BD Totalys™ Datalink
- Remote support through BD Assurity Linc™ to help maximize system uptime
Performance
- Standardized ready-to-use reagents
- <15 minutes hands-on time for run setup
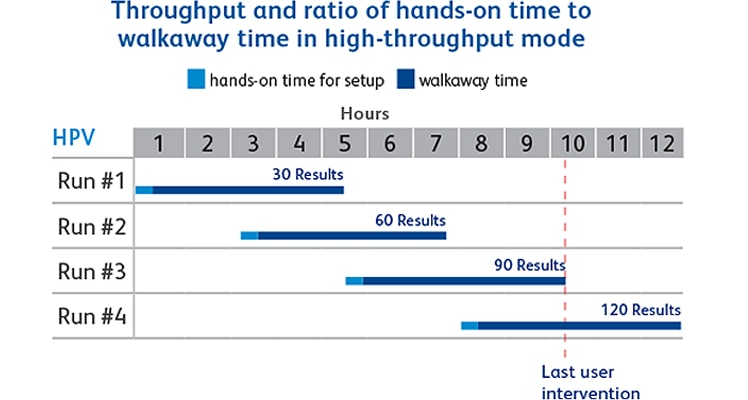
- Up to 30 sample results in 4.5 hours
- 120 results per day. Last user intervention at 9.5 hour in high-throughput mode
- Perform interleaved runs, improving throughput by staggering batches
Comprehensive results
- BD Onclarity™ HPV Assay with extended genotyping is a multiplex real-time PCR assay for the qualitative detection of 14 high-risk HPV types
- On-demand genotyping for individual results of 16, 18, 45, 31, 51 and 52 with pooled results of 33/58, 35/39/68 and 56/59/66
- E6/E7 DNA target
- Utilizes human beta globin as an internal control

The BD SurePath™ Collection Vial offers simple standardized cell collection with positive sample identification and the convenience of being a single source for cytology and molecular testing.
BD Diagnostic Solutions is Now Waters Advanced Diagnostics
BD Diagnostic Solutions and Biosciences have combined with Waters Corporation to create a life sciences and diagnostics leader driven by innovation and a relentless commitment to our customers.
As we begin our new journey as part of Waters, there will be no immediate changes in how we serve you. The way you order your products and services and how you process payment for them will not change. Your points of contact for sales, customer service, technical service and support or clinical services will remain the same.
References
1. BD Viper™ LT System Validation Protocol. VP-7932-01-4193 BD Viper™ LT User Hands-on-time study.
Disclaimer:
This business has been acquired by Waters Corporation (“Waters”). For products referenced on this page, the legal manufacturer remains Becton, Dickinson and Company or one of its affiliates or subsidiaries (“BD”) until all required regulatory transfers are completed. During this interim period, BD maintains full responsibility for all regulatory obligations of the legal manufacturer. Product information provided here is supplied under the regulatory authority of BD. To learn more about the relationship between Waters and BD during this transition period, please see our detailed summary waters.com/bdtransaction.