- Majercik, S. et al. “Strength of tissue attachment to mesh after ventral hernia repair with synthetic composite mesh in a porcine model.” Surg Endos. (2006) 20: 1671-1674.
- Results may not correlate to performance in humans.
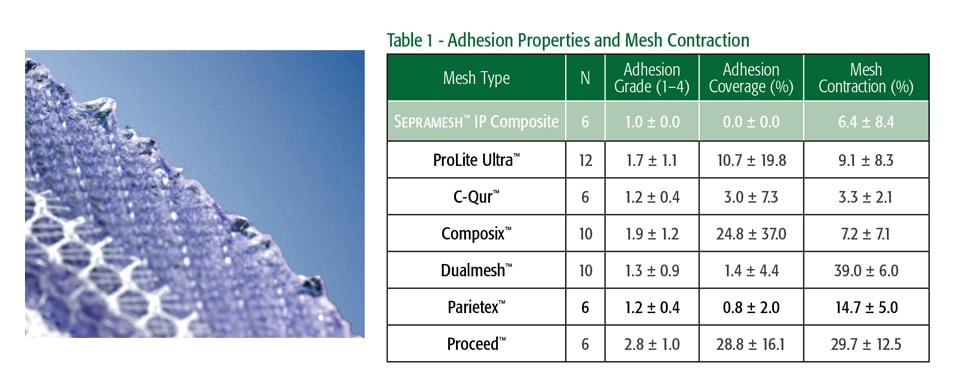
- Pierce, Richard A. MD, PhD, et al. Surgical Innovation. March 2009; 16, 1:45-54.
- Preclinical data on file at C. R. Bard. Results may not correlate to performance in humans.
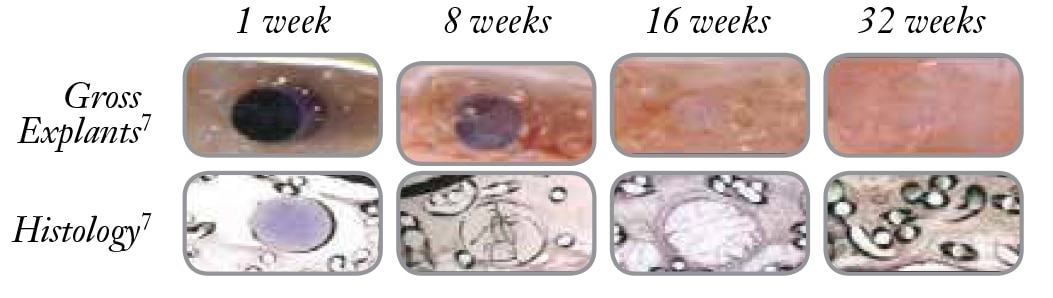
- These images are from a porcine study using the Ventrio™ Hernia Patch which contains the same SorbaFlex™ Memory Technology
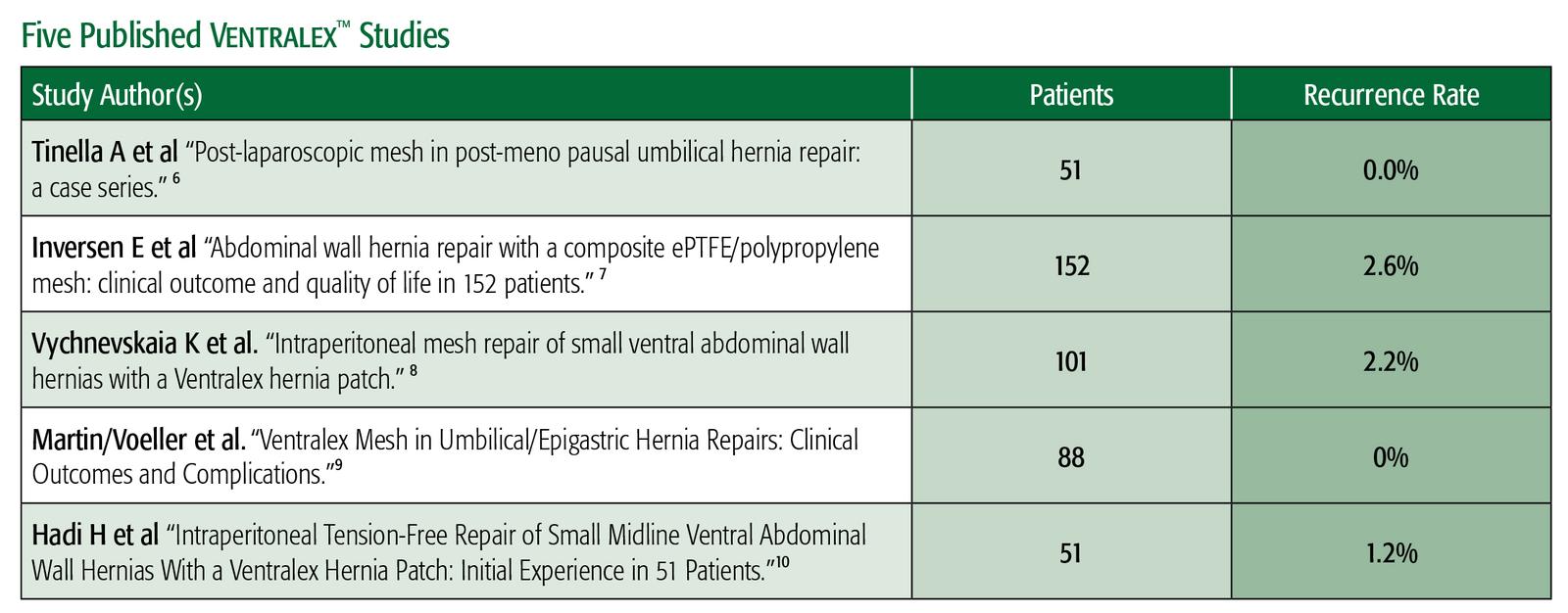
- Tinella A, Malvasi A, Manca C, Alemanno G, Bettocchi S, Benhidjeb T. “Post-laparoscopic mesh in post-menopausal umbilical hernia repair: a case series.” Minim Invasive Ther Allied Technol. 2011 Sep; 20(5):290-5.
- Iversen E, Lykke A, Hensler M, Jorgensen LN. “Abdominal wall hernia repair with a composite ePTFE/polypropylene mesh: clinical outcome and quality of life in 152 patients.” Hernia. 2010 Dec;14(6): 555-60.
- Vychnevskaia K, Mucci-Hennekinne S, Casa C, et al. “Intraperitoneal mesh repair of small ventral abdominal wall hernias with a Ventralex™ Hernia Patch.” Dig Surg. 2010; 27(5): 433-5.
- D.F. Martin, R.F. Williams, T. Mulrooney, and G.R. Voeller. “Ventralex™ Mesh in Umbilical/Epigastric Hernia Repairs: Clinical Outcomes and Complications.” Hernia. 2008 Aug 12(4) 379-83.
- H.I.A. Hadi, A. Maw, S. Sarmah, P. Kumar. “Intraperitoneal Tension-Free Repair of Small Midline Ventral Abdominal Wall Hernias With a Ventralex™ Hernia Patch: Initial Experience in 51 Patients.” Hernia. 2006: 10:409-413.
INDICATIONS
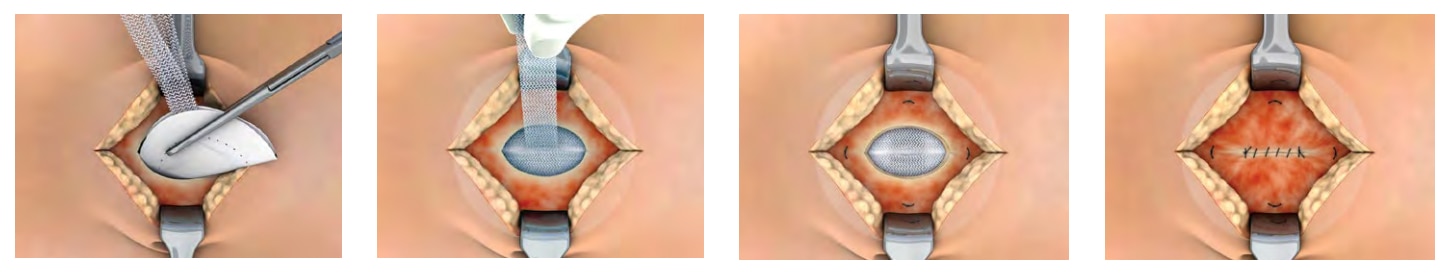
The Ventralex™ ST Hernia Patch is indicated for use in the reinforcement of soft tissue, where weakness exists, in procedures involving soft tissue repair, including repair of hernias and deficiencies caused by trocars.
CONTRAINDICATIONS
Do not use the Ventralex™ ST Hernia Patchin infants or children, whereby future growth will be compromised by the use of such mesh material. Do not use the Ventralex™ ST Hernia Patch for the reconstruction of cardiovascular defects. Literature reports that there may be a possibility for adhesion formation when the polypropylene is placed in contact with the bowel or viscera.
WARNINGS
Do not cut or reshape the Ventralex™ ST Hernia Patch, as this could impact its effectiveness, except for the polypropylene positioning strap. Care should be taken not to cut or nick the SorbaFlex™PDO monofilament during insertion or fixation. If the SorbaFlex™PDO monofilament is cut or damaged, additional complications may include bowel or skin perforation and infection. Follow proper folding techniques for all patches as described in these Instructions for Use as other folding techniques may compromise the SorbaFlex™ PDO monofilament. Ensure proper orientation; the bioresorbable coated side of the prosthesis should be oriented against the bowel or sensitive organs. Do not place the polypropylene mesh side against the bowel. There may be a possibility for adhesion formation when the mesh is placed in direct contact with the bowel or viscera.
ADVERSE REACTIONS
Possible complications include seroma, adhesions, hematomas, inflammation, extrusion, fistula formation, infection, allergic reaction, and recurrence of the hernia or soft tissue defect. If the SorbaFlex™ PDO monofilament is cut or damaged during insertion or fixation, additional complications may include bowel or skin perforation and infection.
Please consult package insert for more detailed safety information and instructions for use.