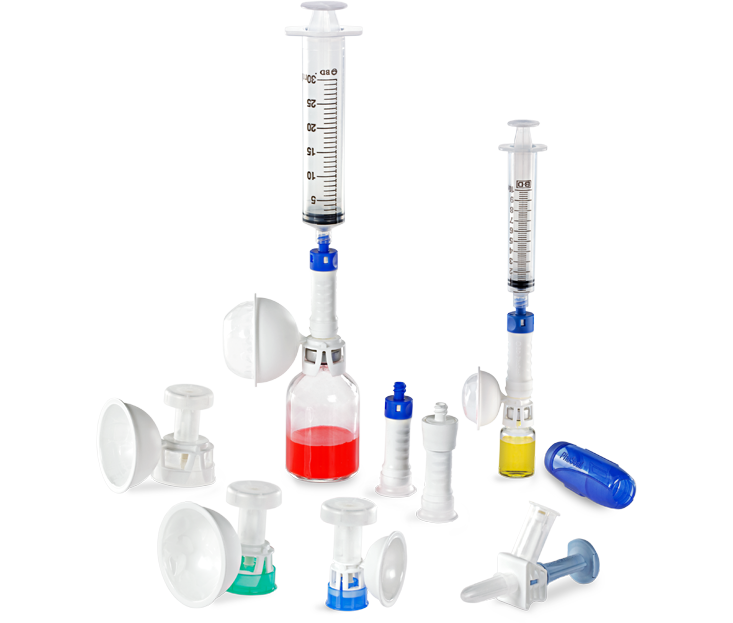
The BD PhaSeal™ System pioneered the category of closed-system drug-transfer devices (CSTDs) to help protect the pharmacists and clinicians who prepare and administer hazardous drugs. Twenty years later, we turned to healthcare professionals like you for feedback and guidance to optimize its every component. The result is the BD PhaSeal™ Optima System—a next-generation, user-tested CSTD solution that advances hazardous drug safety.
BD PhaSeal™ Optima System
The next-generation closed-system drug-transfer Device (CSTD) from BD that advances hazardous drug protection


- Overview
- Why Choose Optima?
- Applications
- Product & Accessories
- EIFU & Resources
Click on each hot spot below to see how we have optimized ease of use, ergonomics and performance.
Notes
* Within an ISO Class V environment following aseptic technique
** 2015
^ The ability to prevent microbial ingress for up to 168 hours should not be interpreted as modifying, extending or superseding a manufacturer's labeling recommendations for the storage and expiration dating of the drug vial. Refer to drug manufacturer's recommendations and USP compounding guidelines for shelf life and sterility information.
References
- Centers for Disease Control and Prevention (CDC). Easy Ergonomics: A Guide to Selecting Non-Powered Hand Tools. https://www.cdc.gov/niosh/docs/2004-164/pdfs/2004-164.pdf. Published August 18, 2004. Accessed September 2, 2020.
- BD Engineering study: Evaluation of PhaSeal Optima residual drug loss performance testing. 2017.
- BD Engineering study: Evaluation of PhaSeal Optima residual drug loss performance testing. 2018.
- Gilbar PJ, Chambers CR, Gilbar EC. Opportunities to significantly reduce expenditure associated with cancer drugs. Future Oncol.2017;13(15):1311-1322.
- McMichael DM, Jefferson DM, Carey ET, et al. Utility of the PhaSeal closed system drug transfer device. Am J Pharm Benefits.2011;3(1):9-16.
- Bendeka [prescribing information], North Wales, Pennsylvania. Teva Pharmaceuticals USA, Inc. 2018.
- Doxil [prescribing information], Bedford, Ohio. ALZA Corporation. 2016.
- Herceptin [prescribing information], South San Francisco, California. Genentech, Inc. 2017.
- Valstar [prescribing information], Chadds Ford, Pennsylvania. Endo Pharmaceuticals, Inc. 2017
- April 2018 ASP pricing file. CMS website. https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Part-B-Drugs/McrPartBDrugAvgSalesPrice/2018ASPFiles.html. Accessed June 8, 2018.
BD-17517 (03/26)
Designed to address your economic concerns
Ease of use
The BD PhaSeal™ Optima System is easy to use, and connects with one step
The BD PhaSeal™ Optima System replaces the push-turn-push connection mechanism of the BD PhaSeal™ System with a one-step straight-push mechanism, making it easier for pharmacists and clinicians to use.
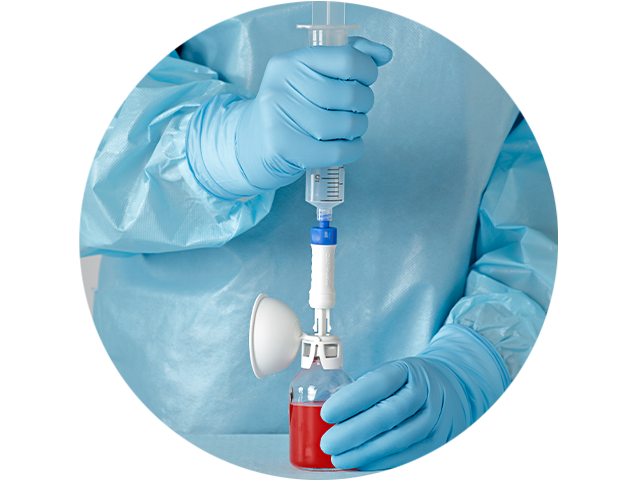
Minimize learning curve
Designed to help minimize the learning curve with:
- Intuitive one-step straight-push connection
- No alignment or orientation needed to connect
Let our trained clinical specialists help you efficiently integrate the BD PhaSeal™ Optima System into the workflow of your facility.
The PhaSeal™ Optima Connect & Go System for Pharmacy and Nursing
The PhaSeal™ Optima Connect & Go System, comprising the PhaSeal™ Optima Spike Set C180-O (515628) and the PhaSeal™ Optima Connecting Set C83-O (515629), is designed to:
- Enable the use of a single line to administer multiple non-hazardous and hazardous drugs.
- Help reduce the need for priming of multiple infusion lines.
- Enable a dry disconnection of mated components between an IV container and infusion set.
The Spike Set is a single-patient use sterile bag access device that enables preparation and administration of non-hazardous and hazardous parenteral drugs when used with devices that have the compatible mating component – the Injector, and/or Connecting Set.
The Connecting Set is a single patient use sterile device that enables administration of non-hazardous and hazardous parenteral drugs when used with devices that have the compatible mating component – the Spike Set and/or Connector.
Ergonomics
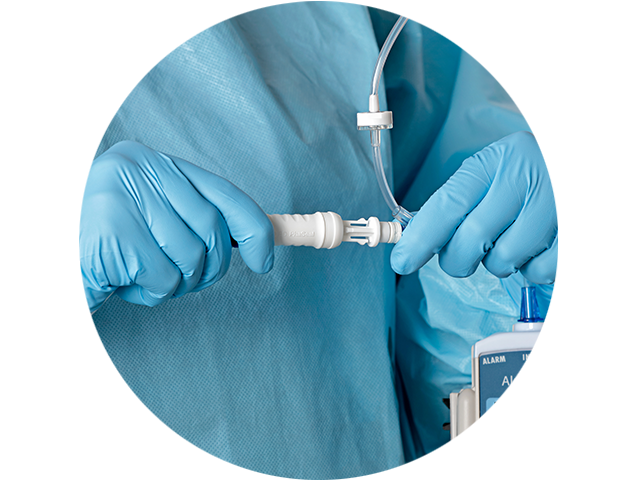
Offer your pharmacists and clinicians an ergonomic CSTD solution
We conducted head-to-head comparisons and asked both longstanding CSTD users and traditional technique users how we could improve our design. The feedback and validation we received guided us to optimize every component of the BD PhaSeal™ Optima System for user comfort.
Align with NIOSH recommendations1
- Uses a power grip and straight motion to connect and disconnect
- Aligns with NIOSH recommendations to avoid pinch grip for hand-held tools1
Our trained clinical specialists can also help train your pharmacists and clinicians in best practices for preparation and administration using the BD PhaSeal™ Optima System.
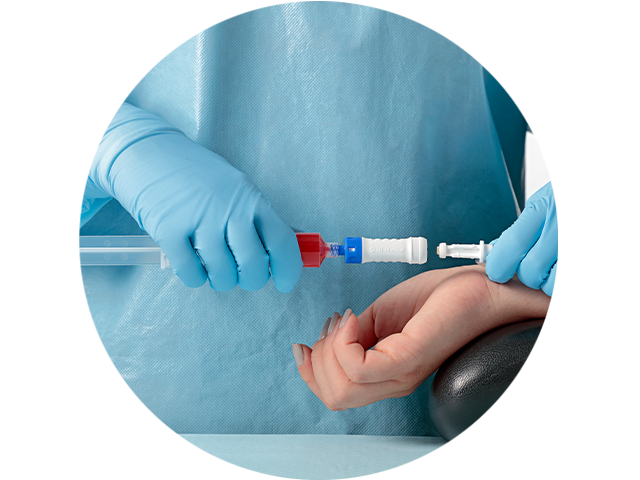
Performance
The BD PhaSeal™ Optima System can help you maximize drug extraction for each vial
In a performance comparison, the BD PhaSeal™ Optima System demonstrated the lowest residual fluid loss of all four CSTDs tested.2,3* Minimizing residual fluid loss with such a CSTD may translate into a reduction in drug waste that can help optimize preparation of HDs and increase cost savings for your facility. 4,5
*Bench test results may not be indicative of clinical results.


Designed to maximize performance
- Prevents microbial ingress* and may maintain drug vial sterility for up to 168 hours and 10 penetrations
- Meets the requirements of the NIOSH IPA draft CSTD-test protocol, a robust test protocol applicable to barrier-type CSTDs*
- Optimized spike-fluid channel and secure vial attachment engineered to common International Organization for Standardization (ISO) standards
- Compatible and connects with ISO-compliant syringes, IV sets, luer fittings and standard-size drug vials
We can share the 'Minimizing residual drug loss' and 'Assessment of microbial ingress' whitepapers with you, and help you calculate potential drug savings for your facility.
*Within an ISO Class V environment following aseptic technique. The ability to prevent microbial ingress for up to 168 hours should not be interpreted as modifying, extending or superseding a manufacturer's labeling recommendations for the storage and expiration dating of the drug vial. Refer to drug manufacturer's recommendations and USP compounding guidelines for shelf life and sterility information.
Safety
Designed to reduce the risk of surface and environmental contamination of hazardous drugs
The BD PhaSeal™ Optima System may help improve healthcare worker safety by reducing the risk of surface contamination to hazardous drugs. The BD PhaSeal™ Optima System incorporates the double membranes and vapor capture mechanisms pioneered by the BD PhaSeal™ System, which has been demonstrated to reduce the risk of hazardous drug exposure.
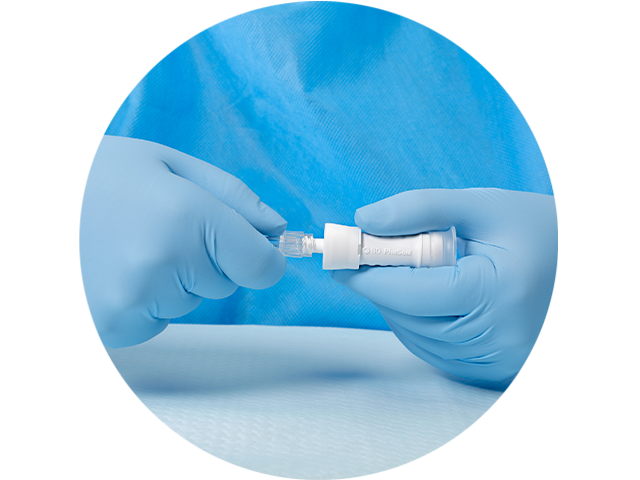
Secure your luer lock connections with the option of the N40-O locking injector
- Locking feature that locks securely to the mated Luer device
- Spinning collar enables rotation of syringe to view scale markings and prevent tangling of IV lines
- May reduce drug leaks, large spills and waste
- Helps prevent leaks in transport to clinical settings

Leakproof and airtight by design
- Spike design prevents vapor leakage when puncturing a vial
- Self-sealing membranes on all components
- Airtight drug transfers in preparation and administration
Let our trained clinical specialists help you seamlessly upgrade your facility to the BD PhaSeal™ Optima System.
Notes
* Within an ISO Class V environment following aseptic technique
** 2015
^ The ability to prevent microbial ingress for up to 168 hours should not be interpreted as modifying, extending or superseding a manufacturer's labeling recommendations for the storage and expiration dating of the drug vial. Refer to drug manufacturer's recommendations and USP compounding guidelines for shelf life and sterility information.
References
- Centers for Disease Control and Prevention (CDC). Easy Ergonomics: A Guide to Selecting Non-Powered Hand Tools. https://www.cdc.gov/niosh/docs/2004-164/pdfs/2004-164.pdf. Published August 18, 2004. Accessed September 2, 2020.
- BD Engineering study: Evaluation of PhaSeal Optima residual drug loss performance testing. 2017.
- BD Engineering study: Evaluation of PhaSeal Optima residual drug loss performance testing. 2018.
- Gilbar PJ, Chambers CR, Gilbar EC. Opportunities to significantly reduce expenditure associated with cancer drugs. Future Oncol.2017;13(15):1311-1322.
- McMichael DM, Jefferson DM, Carey ET, et al. Utility of the PhaSeal closed system drug transfer device. Am J Pharm Benefits.2011;3(1):9-16.
- Bendeka [prescribing information], North Wales, Pennsylvania. Teva Pharmaceuticals USA, Inc. 2018.
- Doxil [prescribing information], Bedford, Ohio. ALZA Corporation. 2016.
- Herceptin [prescribing information], South San Francisco, California. Genentech, Inc. 2017.
- Valstar [prescribing information], Chadds Ford, Pennsylvania. Endo Pharmaceuticals, Inc. 2017
- April 2018 ASP pricing file. CMS website. https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Part-B-Drugs/McrPartBDrugAvgSalesPrice/2018ASPFiles.html. Accessed June 8, 2018.
BD-17517 (03/26)
Easy to use in the preparation and administration of hazardous drugs

See how pharmacists and clinicians can implement the airtight and leakproof BD PhaSeal™ Optima System.
BD PhaSeal™ Optima System instructional video for preparation
BD PhaSeal™ Optima System instructional video for administration

Notes
* Within an ISO Class V environment following aseptic technique
** 2015
^ The ability to prevent microbial ingress for up to 168 hours should not be interpreted as modifying, extending or superseding a manufacturer's labeling recommendations for the storage and expiration dating of the drug vial. Refer to drug manufacturer's recommendations and USP compounding guidelines for shelf life and sterility information.
References
- Centers for Disease Control and Prevention (CDC). Easy Ergonomics: A Guide to Selecting Non-Powered Hand Tools. https://www.cdc.gov/niosh/docs/2004-164/pdfs/2004-164.pdf. Published August 18, 2004. Accessed September 2, 2020.
- BD Engineering study: Evaluation of PhaSeal Optima residual drug loss performance testing. 2017.
- BD Engineering study: Evaluation of PhaSeal Optima residual drug loss performance testing. 2018.
- Gilbar PJ, Chambers CR, Gilbar EC. Opportunities to significantly reduce expenditure associated with cancer drugs. Future Oncol.2017;13(15):1311-1322.
- McMichael DM, Jefferson DM, Carey ET, et al. Utility of the PhaSeal closed system drug transfer device. Am J Pharm Benefits.2011;3(1):9-16.
- Bendeka [prescribing information], North Wales, Pennsylvania. Teva Pharmaceuticals USA, Inc. 2018.
- Doxil [prescribing information], Bedford, Ohio. ALZA Corporation. 2016.
- Herceptin [prescribing information], South San Francisco, California. Genentech, Inc. 2017.
- Valstar [prescribing information], Chadds Ford, Pennsylvania. Endo Pharmaceuticals, Inc. 2017
- April 2018 ASP pricing file. CMS website. https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Part-B-Drugs/McrPartBDrugAvgSalesPrice/2018ASPFiles.html. Accessed June 8, 2018.
BD-17517 (03/26)
-
Protect clinicians and patients from exposure to hazardous drugs with a closed system
-

Get results in less than 10 minutes with the first and only rapid hazardous drug* detection system
Our collection of literature on industries and on our offerings gives you information you can use to continue striving for excellence.
BD PhaSeal™ Optima video for pharmacy
BD PhaSeal™ Optima System instructional video for administration
BD PhaSeal™ Optima System
Connecting BD PhaSeal™ Optima locking Injector with a spin collar instructional video
BD PhaSeal™ Optima video for pharmacy
BD PhaSeal™ Optima System instructional video for administration
BD PhaSeal™ Optima System
Connecting BD PhaSeal™ Optima locking Injector with a spin collar instructional video








