The OptiFix™ Open Absorbable Fixation System was designed for improved ease of use when fixating surgical meshes to the abdominal wall in open ventral hernia procedures by reducing limitations that exist with current laparoscopic straight devices. Combined with Ventrio™ ST, it provides a full system for open ventral hernia repair.
OptiFix™ Open Absorbable Fixation System
Advancing the Open Ventral Hernia Repair Experience

- Overview
- Products & Accessories
- EIFU & Resources
- FAQ
For Open Ventral Hernia Repair
The OptiFix™ Open Absorbable Fastener is made from Poly(D, L-Lactide) and is designed for optimal performance. Fastener features include: Smooth Fastener Head Minimizes the potential for tissue attachment1 Ensures mesh is securely fixated Hollow Core Design Allows tissue ingrowth through the fastener1 Angled Tip Easily penetrates mesh and tissue1 Stabilizers Enhances tissue holding strength Prevents the fastener from backing out
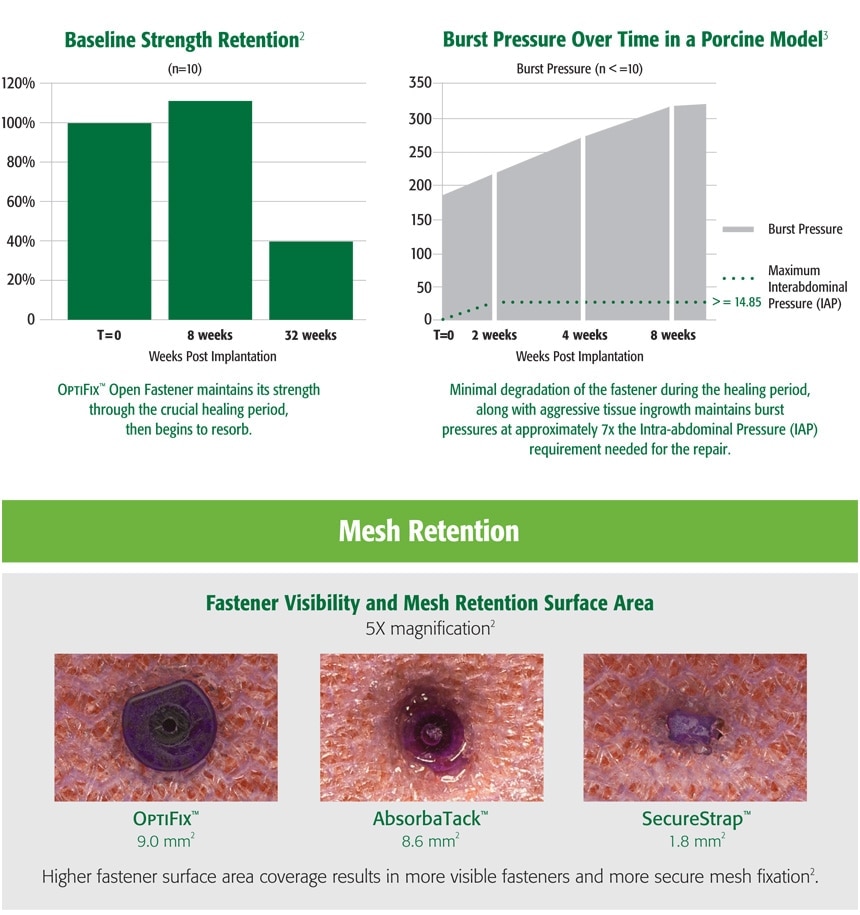
Surgeon Feedback 4 Out of 16 Surgeons surveyed following OptiFix™ Open device testing: 80% favored the overall design of OptiFix™ Open over SecureStrap™ Open, while over 87.5% favored the device over AbsorbaTack™ Short.
1. Preclinical data on file. Results may not correlate to clinical outcomes.
2. C. R. Bard Inc., bench data on file.
3 Preclinical data on file. Results may not correlate to clinical outcomes.
4 Survey of surgeons attending pre-clinical lab.
Results may not correlate to clinical outcomes. Data on file.
OptiFix™ Open Absorbable Fixation System
Indications
The OptiFix™ Open Absorbable Fixation System is indicated for the fixation of surgical mesh to tissues during open surgical procedures, such as hernia repair.
Contraindications
This device is not intended for use except as indicated. 2. Do not use this device where hemostasis cannot be verified visually after application
Contraindications associated with open surgical procedures relative to mesh fixation apply, including but not limited to:
- Fixation of vascular or neural structures
- Fixation of bone and cartilage,
- Situations with insufficient ingrowth of tissue into the mesh over time, which could result in inadequate fixation once the fastener is absorbed.
- Carefully inspect the area in the vicinity of the tissue being fastened to avoid inadvertent penetration of underlying structures such as nerves, vessels, viscera or bone. Use of the OptiFix™ Open Absorbable Fixation System in the close vicinity of such underlying structures is contraindicated. For reference, the length of the fastener below the fastener head is 6.1 mm, the fastener head is another 0.6 mm (total 6.7 mm). This device should not be used in tissues that have a direct anatomic relationship to major vascular structures. This would include the deployment of fasteners in the diaphragm in the vicinity of the pericardium, aorta, or inferior vena cava during diaphragmatic hernia repair.
Warnings
The OptiFix™ Open Absorbable Fixation System is intended for Single Use Only – DO NOT RESTERILIZE. Reuse, reprocessing, resterilization or repackaging may compromise the structural integrity and/or essential material and design characteristics that are critical to the overall performance of the device and may lead to device failure which may result in injury to the patient. Reuse, reprocessing, resterilization or repackaging may also create a risk of contamination of the device and/or cause patient infection or cross infection, including, but not limited to, the transmission of infectious diseases from one patient to another. Contamination of the device may lead to injury, illness or death of the patient or end user. Do not use beyond the expiration date on the package. Prior to use, carefully examine package and product to verify neither is damaged and that all seals are intact. Do not use if the foil pouch or package is damaged or open, or if the center of the temperature indicator on the foil pouch is black. As with any implant material the presence of bacterial contamination may enhance bacterial infectivity. Accepted surgical practice must be followed with respect to drainage and closure of infected or contaminated wounds. Users should be familiar with surgical procedures and techniques involving synthetic absorbable materials before employing OptiFix™ Open Absorbable Fixation System fasteners for wound closure, as the risk of wound dehiscence may vary with the site of application and the material used. The device may not fixate through prosthetics derived from biologic material such as xenografts and allografts. Prosthetic should be evaluated for compatibility prior to use.
Precautions
Please read all instructions before using the OptiFix™ Open Absorbable Fixation System. 2. Only persons having adequate medical training and familiarity with surgical techniques should perform surgical procedures. Consult the medical literature relative to technique, complications and hazards prior to any surgical procedure. 3. Counterpressure should be applied on the target area. Avoid placing hand/finger directly over the area where fastener is being deployed to prevent injury. 4. Use caution when applying the OptiFix™ Open fastener over or in proximity to underlying bone, vessels, nerves, or viscera. The intended fixation site should be assessed to ensure that while the tissue is compressed the total distance from the surface of the tissue to any underlying structures is greater than the length of the OptiFix™ Open fastener. 5. Insertion of fasteners is possible into some collagenous structures such as ligaments and tendons, but is NOT possible directly into bone or cartilage. This may damage the device and result in compromised fixation strength. 6. Care should be taken not to use excessive counterpressure as this may damage the distal tip of the device as well as the mesh and/or tissue. 7. If the device locks and cannot be separated from a fastener that has been deployed into mesh and/or tissue, place a grasper adjacent to the deployed fastener and pull to free the device. If needed, you may use laparoscopic scissors to cut below the fastener head. The remaining portion of the fastener stem left in the mesh can be removed with graspers. The device should then be discarded and a new device should be used. 8. If the fastener does not deploy properly, remove the device from the patient and test the device in gauze to ensure proper fastener deployment. Once proper fastener deployment is confirmed, the device may be reinserted into the patient.
Adverse Reactions
Adverse reactions and potential complications associated with fixation devices such as the OptiFix™ Open Absorbable Fixation System may include, but are not limited to the following: hemorrhage; pain, edema and erythema at wound site; allergic reaction to Poly(D, L)-lactide; infection/septicemia; hernia recurrence/wound dehiscence.
Please consult product labels and inserts for any indications, contraindications, hazards, warnings, precautions and instructions for use.
1. Preclinical data on file. Results may not correlate to clinical outcomes.
2. C. R. Bard Inc., bench data on file.
3 Preclinical data on file. Results may not correlate to clinical outcomes.
4 Survey of surgeons attending pre-clinical lab.
Results may not correlate to clinical outcomes. Data on file.
OptiFix™ Open Absorbable Fixation System
Indications
The OptiFix™ Open Absorbable Fixation System is indicated for the fixation of surgical mesh to tissues during open surgical procedures, such as hernia repair.
Contraindications
This device is not intended for use except as indicated. 2. Do not use this device where hemostasis cannot be verified visually after application
Contraindications associated with open surgical procedures relative to mesh fixation apply, including but not limited to:
- Fixation of vascular or neural structures
- Fixation of bone and cartilage,
- Situations with insufficient ingrowth of tissue into the mesh over time, which could result in inadequate fixation once the fastener is absorbed.
- Carefully inspect the area in the vicinity of the tissue being fastened to avoid inadvertent penetration of underlying structures such as nerves, vessels, viscera or bone. Use of the OptiFix™ Open Absorbable Fixation System in the close vicinity of such underlying structures is contraindicated. For reference, the length of the fastener below the fastener head is 6.1 mm, the fastener head is another 0.6 mm (total 6.7 mm). This device should not be used in tissues that have a direct anatomic relationship to major vascular structures. This would include the deployment of fasteners in the diaphragm in the vicinity of the pericardium, aorta, or inferior vena cava during diaphragmatic hernia repair.
Warnings
The OptiFix™ Open Absorbable Fixation System is intended for Single Use Only – DO NOT RESTERILIZE. Reuse, reprocessing, resterilization or repackaging may compromise the structural integrity and/or essential material and design characteristics that are critical to the overall performance of the device and may lead to device failure which may result in injury to the patient. Reuse, reprocessing, resterilization or repackaging may also create a risk of contamination of the device and/or cause patient infection or cross infection, including, but not limited to, the transmission of infectious diseases from one patient to another. Contamination of the device may lead to injury, illness or death of the patient or end user. Do not use beyond the expiration date on the package. Prior to use, carefully examine package and product to verify neither is damaged and that all seals are intact. Do not use if the foil pouch or package is damaged or open, or if the center of the temperature indicator on the foil pouch is black. As with any implant material the presence of bacterial contamination may enhance bacterial infectivity. Accepted surgical practice must be followed with respect to drainage and closure of infected or contaminated wounds. Users should be familiar with surgical procedures and techniques involving synthetic absorbable materials before employing OptiFix™ Open Absorbable Fixation System fasteners for wound closure, as the risk of wound dehiscence may vary with the site of application and the material used. The device may not fixate through prosthetics derived from biologic material such as xenografts and allografts. Prosthetic should be evaluated for compatibility prior to use.
Precautions
Please read all instructions before using the OptiFix™ Open Absorbable Fixation System. 2. Only persons having adequate medical training and familiarity with surgical techniques should perform surgical procedures. Consult the medical literature relative to technique, complications and hazards prior to any surgical procedure. 3. Counterpressure should be applied on the target area. Avoid placing hand/finger directly over the area where fastener is being deployed to prevent injury. 4. Use caution when applying the OptiFix™ Open fastener over or in proximity to underlying bone, vessels, nerves, or viscera. The intended fixation site should be assessed to ensure that while the tissue is compressed the total distance from the surface of the tissue to any underlying structures is greater than the length of the OptiFix™ Open fastener. 5. Insertion of fasteners is possible into some collagenous structures such as ligaments and tendons, but is NOT possible directly into bone or cartilage. This may damage the device and result in compromised fixation strength. 6. Care should be taken not to use excessive counterpressure as this may damage the distal tip of the device as well as the mesh and/or tissue. 7. If the device locks and cannot be separated from a fastener that has been deployed into mesh and/or tissue, place a grasper adjacent to the deployed fastener and pull to free the device. If needed, you may use laparoscopic scissors to cut below the fastener head. The remaining portion of the fastener stem left in the mesh can be removed with graspers. The device should then be discarded and a new device should be used. 8. If the fastener does not deploy properly, remove the device from the patient and test the device in gauze to ensure proper fastener deployment. Once proper fastener deployment is confirmed, the device may be reinserted into the patient.
Adverse Reactions
Adverse reactions and potential complications associated with fixation devices such as the OptiFix™ Open Absorbable Fixation System may include, but are not limited to the following: hemorrhage; pain, edema and erythema at wound site; allergic reaction to Poly(D, L)-lactide; infection/septicemia; hernia recurrence/wound dehiscence.
Please consult product labels and inserts for any indications, contraindications, hazards, warnings, precautions and instructions for use.
1. Preclinical data on file. Results may not correlate to clinical outcomes.
2. C. R. Bard Inc., bench data on file.
3 Preclinical data on file. Results may not correlate to clinical outcomes.
4 Survey of surgeons attending pre-clinical lab.
Results may not correlate to clinical outcomes. Data on file.
OptiFix™ Open Absorbable Fixation System
Indications
The OptiFix™ Open Absorbable Fixation System is indicated for the fixation of surgical mesh to tissues during open surgical procedures, such as hernia repair.
Contraindications
This device is not intended for use except as indicated. 2. Do not use this device where hemostasis cannot be verified visually after application
Contraindications associated with open surgical procedures relative to mesh fixation apply, including but not limited to:
- Fixation of vascular or neural structures
- Fixation of bone and cartilage,
- Situations with insufficient ingrowth of tissue into the mesh over time, which could result in inadequate fixation once the fastener is absorbed.
- Carefully inspect the area in the vicinity of the tissue being fastened to avoid inadvertent penetration of underlying structures such as nerves, vessels, viscera or bone. Use of the OptiFix™ Open Absorbable Fixation System in the close vicinity of such underlying structures is contraindicated. For reference, the length of the fastener below the fastener head is 6.1 mm, the fastener head is another 0.6 mm (total 6.7 mm). This device should not be used in tissues that have a direct anatomic relationship to major vascular structures. This would include the deployment of fasteners in the diaphragm in the vicinity of the pericardium, aorta, or inferior vena cava during diaphragmatic hernia repair.
Warnings
The OptiFix™ Open Absorbable Fixation System is intended for Single Use Only – DO NOT RESTERILIZE. Reuse, reprocessing, resterilization or repackaging may compromise the structural integrity and/or essential material and design characteristics that are critical to the overall performance of the device and may lead to device failure which may result in injury to the patient. Reuse, reprocessing, resterilization or repackaging may also create a risk of contamination of the device and/or cause patient infection or cross infection, including, but not limited to, the transmission of infectious diseases from one patient to another. Contamination of the device may lead to injury, illness or death of the patient or end user. Do not use beyond the expiration date on the package. Prior to use, carefully examine package and product to verify neither is damaged and that all seals are intact. Do not use if the foil pouch or package is damaged or open, or if the center of the temperature indicator on the foil pouch is black. As with any implant material the presence of bacterial contamination may enhance bacterial infectivity. Accepted surgical practice must be followed with respect to drainage and closure of infected or contaminated wounds. Users should be familiar with surgical procedures and techniques involving synthetic absorbable materials before employing OptiFix™ Open Absorbable Fixation System fasteners for wound closure, as the risk of wound dehiscence may vary with the site of application and the material used. The device may not fixate through prosthetics derived from biologic material such as xenografts and allografts. Prosthetic should be evaluated for compatibility prior to use.
Precautions
Please read all instructions before using the OptiFix™ Open Absorbable Fixation System. 2. Only persons having adequate medical training and familiarity with surgical techniques should perform surgical procedures. Consult the medical literature relative to technique, complications and hazards prior to any surgical procedure. 3. Counterpressure should be applied on the target area. Avoid placing hand/finger directly over the area where fastener is being deployed to prevent injury. 4. Use caution when applying the OptiFix™ Open fastener over or in proximity to underlying bone, vessels, nerves, or viscera. The intended fixation site should be assessed to ensure that while the tissue is compressed the total distance from the surface of the tissue to any underlying structures is greater than the length of the OptiFix™ Open fastener. 5. Insertion of fasteners is possible into some collagenous structures such as ligaments and tendons, but is NOT possible directly into bone or cartilage. This may damage the device and result in compromised fixation strength. 6. Care should be taken not to use excessive counterpressure as this may damage the distal tip of the device as well as the mesh and/or tissue. 7. If the device locks and cannot be separated from a fastener that has been deployed into mesh and/or tissue, place a grasper adjacent to the deployed fastener and pull to free the device. If needed, you may use laparoscopic scissors to cut below the fastener head. The remaining portion of the fastener stem left in the mesh can be removed with graspers. The device should then be discarded and a new device should be used. 8. If the fastener does not deploy properly, remove the device from the patient and test the device in gauze to ensure proper fastener deployment. Once proper fastener deployment is confirmed, the device may be reinserted into the patient.
Adverse Reactions
Adverse reactions and potential complications associated with fixation devices such as the OptiFix™ Open Absorbable Fixation System may include, but are not limited to the following: hemorrhage; pain, edema and erythema at wound site; allergic reaction to Poly(D, L)-lactide; infection/septicemia; hernia recurrence/wound dehiscence.
Please consult product labels and inserts for any indications, contraindications, hazards, warnings, precautions and instructions for use.