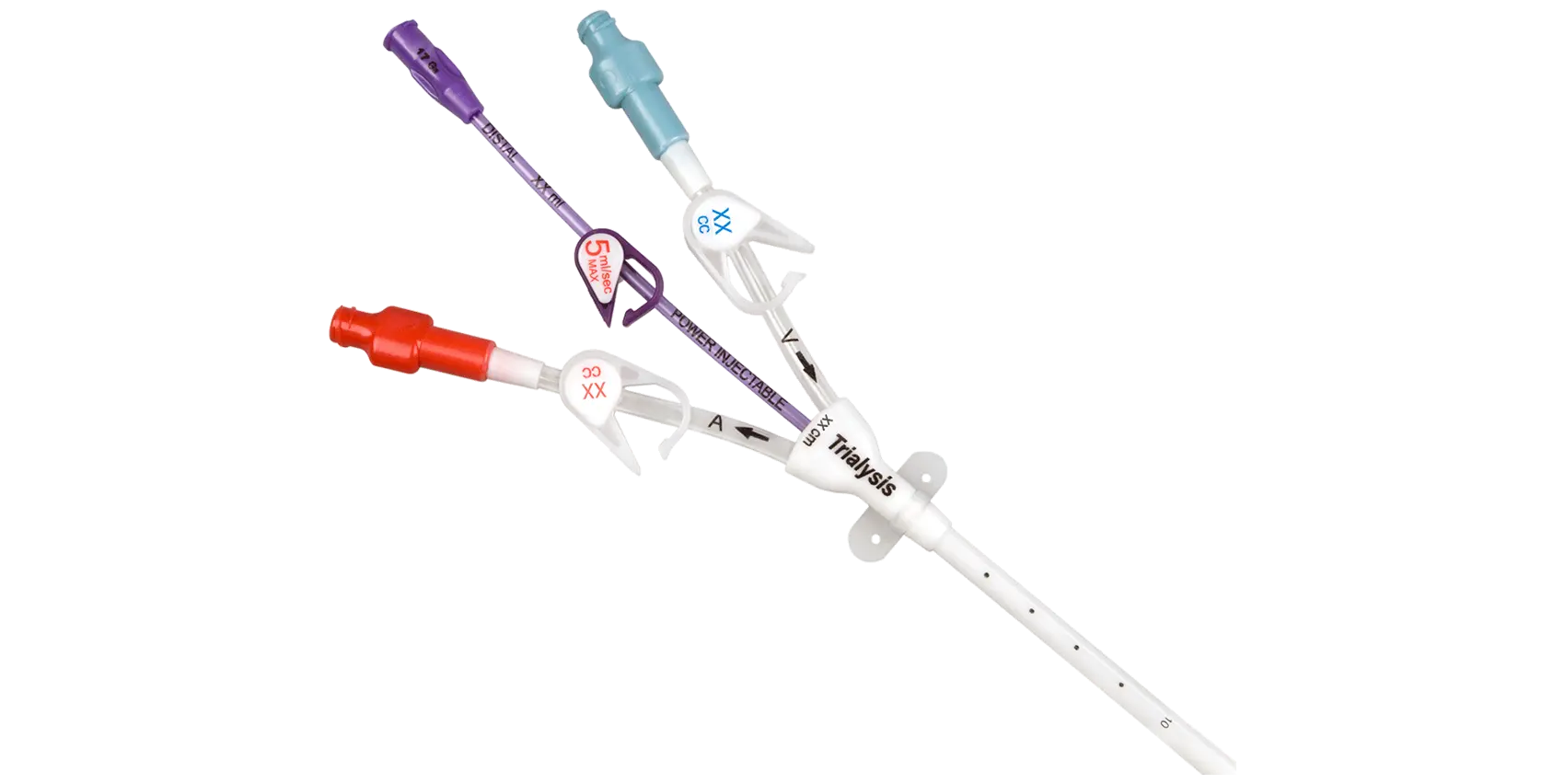
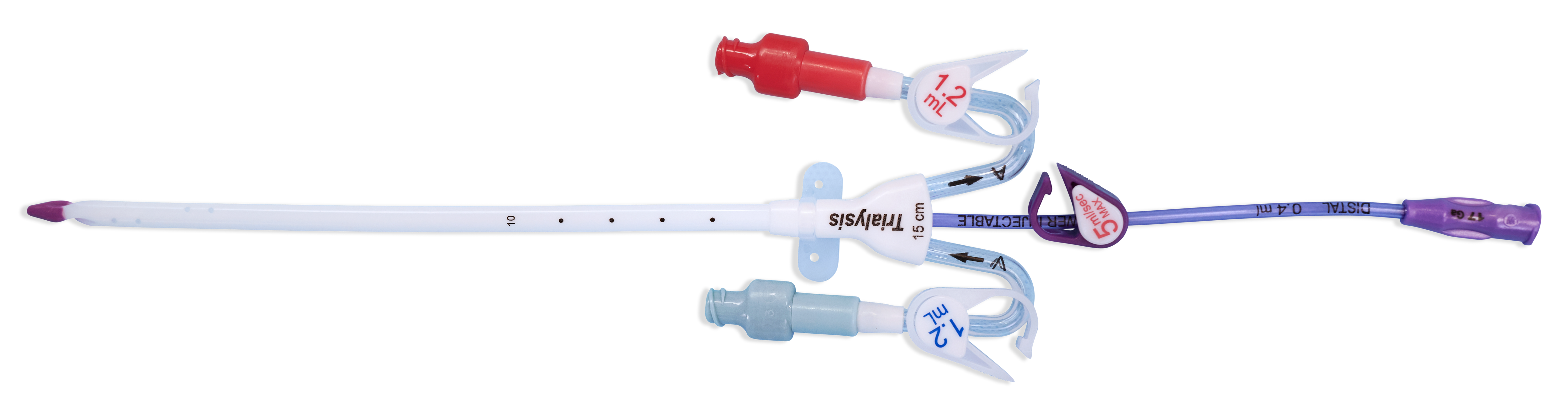
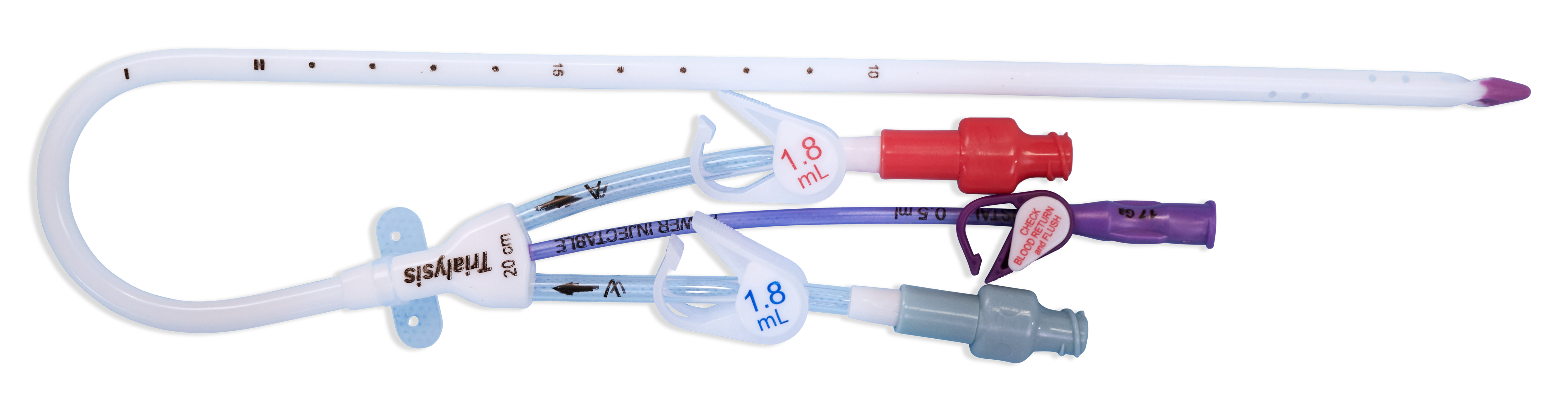
- Demonstrated a reliable flow rate up to 400 mL/min and recirculation of 2% on average through simulated testing*
- Enables clinicians to facilitate compliance with federal regulations and guidelines
*As demonstrated through simulated testing. Results may not be indicative of actual clinical performance. Different tests may yield different results.