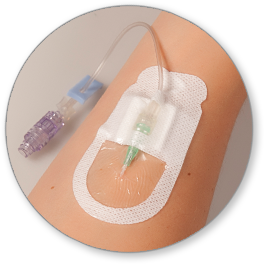
The Securis™ Stabilization Device was designed to secure non-winged PIVC catheters and minimize skin irritation.
true

- Overview
- Products & Accessories
- eIFU & Resources
Highlights
true
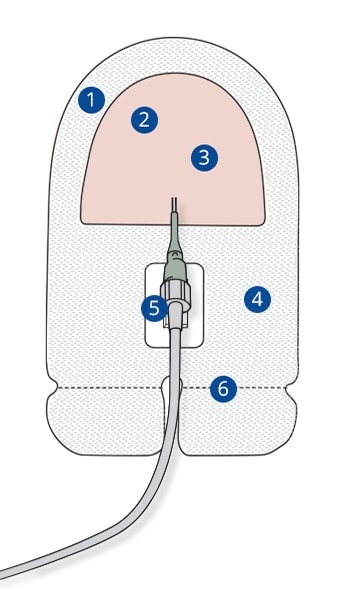
Securis™ Stabilization Device
- Reinforced Border: Prevent the dressing from rolling up using a reinforced border design.
- Transparent Film: Support vapor and oxygen exchange.
- Insertion Site Window: Allow continuous site observation as recommended by the CDC and Infusion Nursing Standards of Practice.
- Transparent Dressing: Provide a viral barrier while the dressing remains intact without leakage.
- Luer Window: Allow continuous observation of the Luer nut and the catheter hub connection.
- Perforation: Aids in device removal.
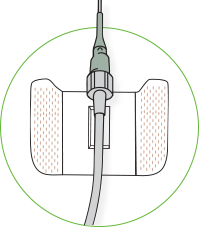
The comfort pad is designed to:
- Provide secure non-winged PIVC catheters and minimize skin irritation
- Create a cushion between the Luer nut and the patient’s skin1
- Accommodate non-winged PIVC catheters
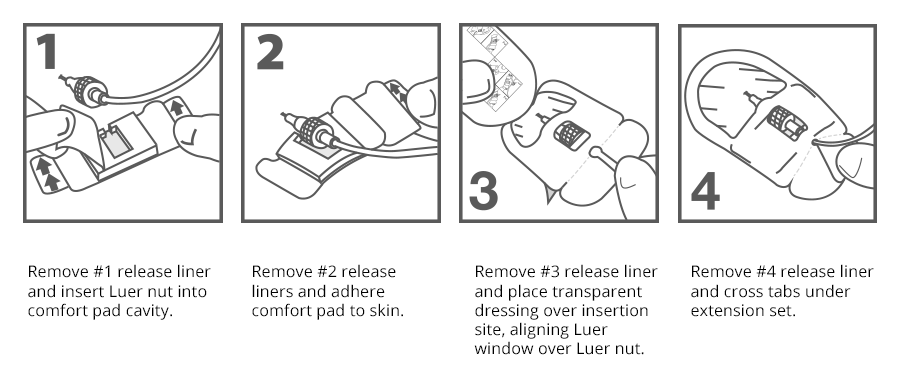
Application Technique
Prep the targeted stabilization site with alcohol or per hospital policy to degrease skin. Allow to dry completely.
1As demonstrated through simulated testing. Results may not be indicative of actual clinical performance. Different test may yield different results.
BD-83750 (05/23)
BD-83750 (05/23)
true
true
BD-83750 (05/23)
true