-
SenoMark™ Breast Tissue Marker
O shape made of Titanium - Compatible with ATEC™ Probe - 9G
SKU/REF smtsu9g
-
SenoMark™ Breast Tissue Marker
O shape made of Titanium - Compatible with Classic Mammotome™ Probe™ - 11G
SKU/REF smtmt11g
-
SenoMark™ Breast Tissue Marker
O shape made of Titanium - Compatible with EnCor™ Probe - 10G
SKU/REF smtec10g
-
SenoMark™ Breast Tissue Marker
X shape made of Titanium - Compatible with Standard Eviva™ Probe™ - 9G
SKU/REF smse9gt
-
SenoMark™ Breast Tissue Marker
X shape made of Titanium - Compatible with ATEC™ Probe - 9G
SKU/REF smrsu9gt
-
SenoMark™ Breast Tissue Marker
S shape made of Titanium - Compatible with ATEC™ Probe - 12G
SKU/REF smrsu12gt
-
SenoMark™ Breast Tissue Marker
M shape made of 316L Stainless Steel - Compatible with EnCor™ Probe - 7G
SKU/REF smec7gss
-
SenoMark™ Breast Tissue Marker
Omega shape made of 316L Stainless Steel - Compatible with EnCor™ Probe - 12G
SKU/REF smec12gss
-
SenoMark™ Breast Tissue Marker
M shape made of 316L Stainless Steel - Compatible with EnCor™ Probe - 10G
SKU/REF smec10gss


- Overview
- Products & Accessories
- EIFU & Resources
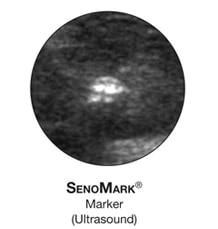
The SenoMark™ Breast Tissue Marker deploys 3 polyglycolic acid pads with one radiopaque marker. The pads are visible via ultrasound for approximately 3 weeks.
Designed for accuracy and visibility
Ultrasound visibility

Marker Shapes that Celebrate Her
With each purchase of the Heart, Venus or Ring breast tissue marker shapes, BD will contribute $1 to the American Cancer Society™ in honor of breast cancer patients.**
* Applies to specially marked BD breast tissue marker products purchased from BD. The American Cancer Society™ does not endorse any service or product. Visit bd.com/icoh for details. Valid through September 30.
Please consult product labels and inserts for indications, contraindications, hazards, warnings, precautions and directions for use.
BD-110490