-
Elyra™ Thulium Laser Fiber: Single Use 272 μm Ball Tip
The Elyra™ Thulium Laser Fiber is a fiber optic laser energy delivery device capable of delivering thulium radiation energy.
SKU/REF 7082327
-
Elyra™ Thulium Laser Fiber: Single Use 200 μm Ball Tip
The Elyra™ Thulium Laser Fiber is a fiber optic laser energy delivery device capable of delivering thulium radiation energy.
SKU/REF 7082320
-
Elyra™ Thulium Laser Fiber: Single Use 150 μm Ball Tip
The Elyra™ Thulium Laser Fiber is a fiber optic laser energy delivery device capable of delivering thulium radiation energy.
SKU/REF 7082315
-
Elyra™ Thulium Laser Fiber: Single Use 940 μm Standard Tip
The Elyra™ Thulium Laser Fiber is a fiber optic laser energy delivery device capable of delivering thulium radiation energy.
SKU/REF 7082194
-
Elyra™ Thulium Laser Fiber: Single Use 550 μm Standard Tip
The Elyra™ Thulium Laser Fiber is a fiber optic laser energy delivery device capable of delivering thulium radiation energy.
SKU/REF 7082155
-
Elyra™ Thulium Laser Fiber: Single Use 365 μm Standard Tip
The Elyra™ Thulium Laser Fiber is a fiber optic laser energy delivery device capable of delivering thulium radiation energy.
SKU/REF 7082136
-
Elyra™ Thulium Laser Fiber: Single Use 272 μm Standard Tip
The Elyra™ Thulium Laser Fiber is a fiber optic laser energy delivery device capable of delivering thulium radiation energy.
SKU/REF 7082127
-
Elyra™ Thulium Laser Fiber: Single Use 200 μm Standard Tip
The Elyra™ Thulium Laser Fiber is a fiber optic laser energy delivery device capable of delivering thulium radiation energy.
SKU/REF 7082120
-
Elyra™ Thulium Laser Fiber: Single Use 150 μm Standard Tip
The Elyra™ Thulium Laser Fiber is a fiber optic laser energy delivery device capable of delivering thulium radiation energy.
SKU/REF 7082115
-
Elyra™ Plus Thulium Fiber Laser System
The Elyra™ Plus Thulium Fiber Laser (TFL) System is a thulium fiber laser which delivers a pulsed beam of near-infrared light through the active media of a delivery fiber.
SKU/REF 7082002
-
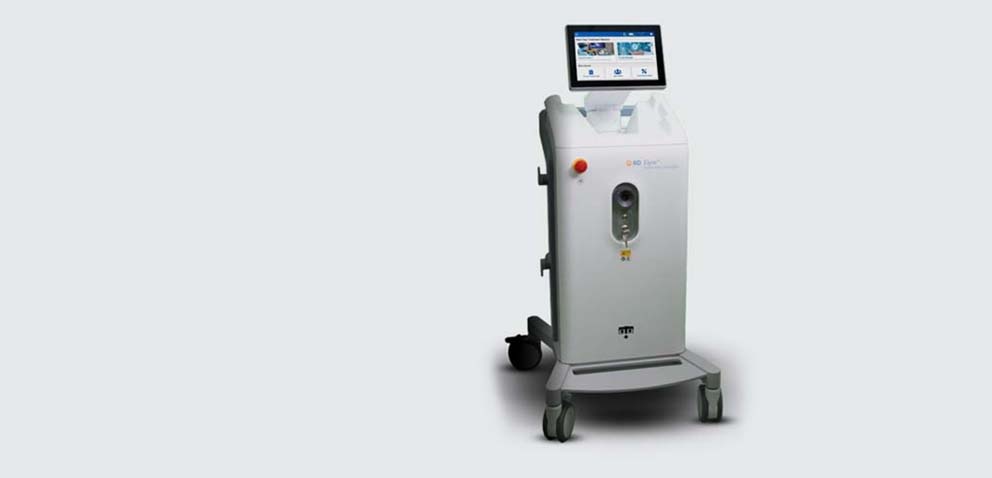
Elyra™ Thulium Fiber Laser System
The Elyra™ Thulium Fiber Laser (TFL) System is a thulium fiber laser which delivers a pulsed beam of near-infrared light through the active media of a delivery fiber.
SKU/REF 7082001
Elyra™ Thulium Fiber Laser System
Own your procedure with the Elyra™ Thulium Fiber Laser System

- Overview
- Products & Accessories
- EIFU & Resources
Putting patients first
Holmium:YAG (Ho:YAG) lasers have long been the standard in urology, but Thulium Fiber Laser (TFL) technology is advancing stone lithotripsy and soft tissue applications with greater efficiency, precision, and cost savings1,2
By streamlining procedures and reducing OR time, TFL gives you back what you need most: time to focus on your patients.1
TFL: A innovation in laser lithotripsy
-

Effective stone dusting
TFL systems are able to dust more effectively and are more procedurally efficient in lithotripsy than Ho:YAG laser systems.1
-

Reduced stone migration
Studies show that when treating urolithiasis, stone migration was significantly reduced with TFL systems compared to Ho:YAG laser systems.1
-

Quieter design
TFL is quieter than traditional Ho:YAG systems due to air-cooling, improving OR conditions.3
-

Standard power compatibility
Does not require a specialty outlet, making OR installation and integration seamless.
With these advantages, is TFL the missing piece to your practice?
With your team and your patients in mind, the Elyra™ Thulium Fiber Laser System is designed to be easy to use and understand. It delivers:
• System usability – designed with a user interface that is easy to understand to streamline workflows.
• User flexibility – with customizable settings, quick start-up, and wireless footswitch.*
• Effective hemostasis capabilities.4
• Standard power consumption – does not require a specialty outlet.
• Quieter operation – quieter than traditional Ho:YAG systems due to air-cooling.
• A system designed with advanced user safety features - including anatomical power notifications.
• Integrated storage for accessories, cords, and footswitch.
• Compatibility with a wide range of BD fibers.
• Trusted BD partnership – With 90+ years in urology, BD provides dedicated support, training, and innovation to help you stay ahead.
The Elyra™ TFL System is designed to be easy to use and understand, with customized presets and portability to seamlessly integrate into your OR.
There’s an Elyra™ TFL System configuration designed for you. Choose the Elyra™ TFL System or Elyra™ Plus TFL System based on your needs—both part of the comprehensive BD Stone Management portfolio.
More than a laser.
A partnership built for your success.
- Seamless compatibility – Elevates BD's Endourology product portfolio, ensuring efficiency and value.
- One trusted partner – Unified support, training, and service from the BD team you already know.
- Future-proof your practice – Equip your OR with our latest innovation in laser lithotripsy and soft tissue treatment.
With BD, you’re not just buying a laser—you’re building the future of urological care.
*Only compatible with the Elyra™ Plus TFL System
- Tang, X., Wu, S., Li, Z. et al. Comparison of Thulium Fiber Laser versus Holmium laser in ureteroscopic lithotripsy: a Meta-analysis and systematic review. BMC Urol 24, 44 (2024). https://doi.org/10.1186/s12894-024-01419-6
- Taratkin M, Kovalenko A, Laukhtina E, et al. Ex vivo study of Ho:YAG and thulium fiber lasers for soft tissue surgery: which laser for which case?. Lasers Med Sci. 2022;37(1):149-154. doi:10.1007/s10103-020-03189-7
- Moore, J., Chavez, A., Narang, G. et al. Operating room noise hazards during laser lithotripsy: a comparison between the thulium fiber and holmium laser platforms. World J Urol 40, 801–805 (2022). https://doi.org/10.1007/s00345-021-03897-x
- Based upon preclinical research results (N = 15 incisions). Averaged results measured at 0, 3, and 10 minutes. Preclinical research results are not indicative of clinical performance or outcomes. Data on file.
Specifications subject to change without notice.
For the latest information, always check the “Instructions for Use” that comes packaged with the product. If you are a consumer seeking more information, please consult your healthcare provider.
Indications for Use:
Elyra™ and Elyra™ Plus TFL System (Laser, Fibers, and Accessories) are intended for incision, excision, resection, ablation, coagulation and hemostasis of soft tissue, with or without an endoscope, in the following indications: urology and lithotripsy.
Urology: Condylomas. Urethral strictures. Lesions of external genitalia. Bladder neck incisions (BNI). Ablation and resection of bladder tumors, urethral tumors, and ureteral tumors. Endoscopic fragmentation of urethral, ureteral, bladder, and renal calculi. Treatment of distal impacted fragments remaining in the ureters following lithotripsy.
Lithotripsy and Percutaneous Urinary Lithotripsy: Endoscopic fragmentation of urethral, ureteral, bladder and renal calculi including cystine, calcium oxalate monohydrate and calcium oxalate dihydrate stones. Endoscopic fragmentation of calculi. Treatment of distal impacted fragments of steinstrasse when guidewire cannot be passed.
In addition, the Elyra™ Plus TFL System (Laser, Fibers, and Accessories) is intended for vaporization of soft tissue, with or without an endoscope, in the following indication: urology.
Urology: Ablation of Benign Prostatic Hyperplasia (Hypertrophy) [BPH]. Laser Resection of the Prostate (LRP). Laser Enucleation of the Prostate (LEP). Laser Ablation of the Prostate (LAP). Transurethral Incision of the Prostate (TUIP).
General Laser Contraindications: Not for treatment of patients for whom endoscopic procedures are contraindicated, whose general medical condition contraindicates surgical intervention, or patients who are intolerant of anesthesia. Not for treatment of patients with septic peritonitis, septic shock, or those who are diagnosed with prostate cancer. Not for resection or excision of large, highly vascularized organs. Tissue (especially the tumor) is calcified. Difficult access to the treatment site in case of: Skeletal anomalies, Renal anomalies, Pleural interposition.
General Urology Contraindications: Acute proneness to bleeding, anticoagulation therapy. Hemostasis of vessels with diameter greater than 2 mm. Untreated infections of the urinary passages.
Please refer to the Elyra™/Elyra™ Plus User Manual for additional contraindications.
Important Safety Information: Improper use of the device or use of a damaged device may result in severe eye, skin or tissue damage, accidental laser exposure to the treatment room personnel or patient which may result in severe thermal injuries or burns to the patient or user, melting or burning of surgical gowns or protective patient drapes, or may cause fire in the treatment room. The laser system is a surgical device that should be operated only by a physician who had training in this laser system and in laser surgery through courses, mentorships, and under the guidance of another physician knowledgeable in laser treatment. Any unauthorized use of the laser system may expose the operator and/or the patient to potential high energy laser radiations or electrical power. When beginning a procedure, start at the lowest setting on the laser system to avoid unexpected soft tissue damage or complication to the patient. To minimize risk of thermal damage to surrounding tissues, continuous irrigation should be used throughout the procedure. Use of the laser system outside of its intended use may cause serious injury to the patient, to the user, or may cause severe damage to the system. Use caution when using the laser system, laser beam can ignite flammable materials within the operating environment. Laser energy may damage guidewires, baskets, and/or other delivery devices during use. The resulting overheating or damage of these devices may cause tissue damage and lead to secondary stricture or rupture in the affected area. Only place the system in READY mode when the laser delivery fiber is at the intended treatment site. The fiber should extend at least 8 mm beyond the scope. The condition of the delivery fiber can be assessed by assessing circularity of the aiming beam profile. Use of damaged laser fiber may result in serious injury to patient. If tissue appears to be stuck to the fiber tip, carefully remove the tip from the tissue and control for hemorrhage and/or tissue lesion. Immediate action might have to be taken. In the event of a detached tip, it may be visually located through an appropriate scope and removed using forceps. Irrigate the area thoroughly to remove any traces of the tip material. Patients who have recently used blood thinner may be at greater risk of uncontrollable bleeding or hematuria during treatment. Treatment of blood vessels of less than 1 mm may cause hemostasis. Before beginning laser delivery, confirm patient is wearing appropriate laser safety eyewear. Increased laser exposure time will result in a deeper and wider zone of tissue necrosis. As with any conventional surgery, discontinue laser treatment immediately if the patient develops any cardiopulmonary problems. The laser system emits class 4 laser radiation when in use. Direct or indirect contact with the laser radiation may cause severe injury to eyes or body. Laser plume may contain toxic gases and vapors, viable tissue particulates, and viruses. Apply properly ventilation, wear appropriate personal PPEs, and apply cleaning techniques to minimize the exposure to the contaminants.
Please refer to the Elyra™/Elyra™ Plus User Manual for additional warnings.
Elyra is a trademark of Becton, Dickinson and Company or its affiliates.


