- Alert
- Products & Accessories
- EIFU & Resources
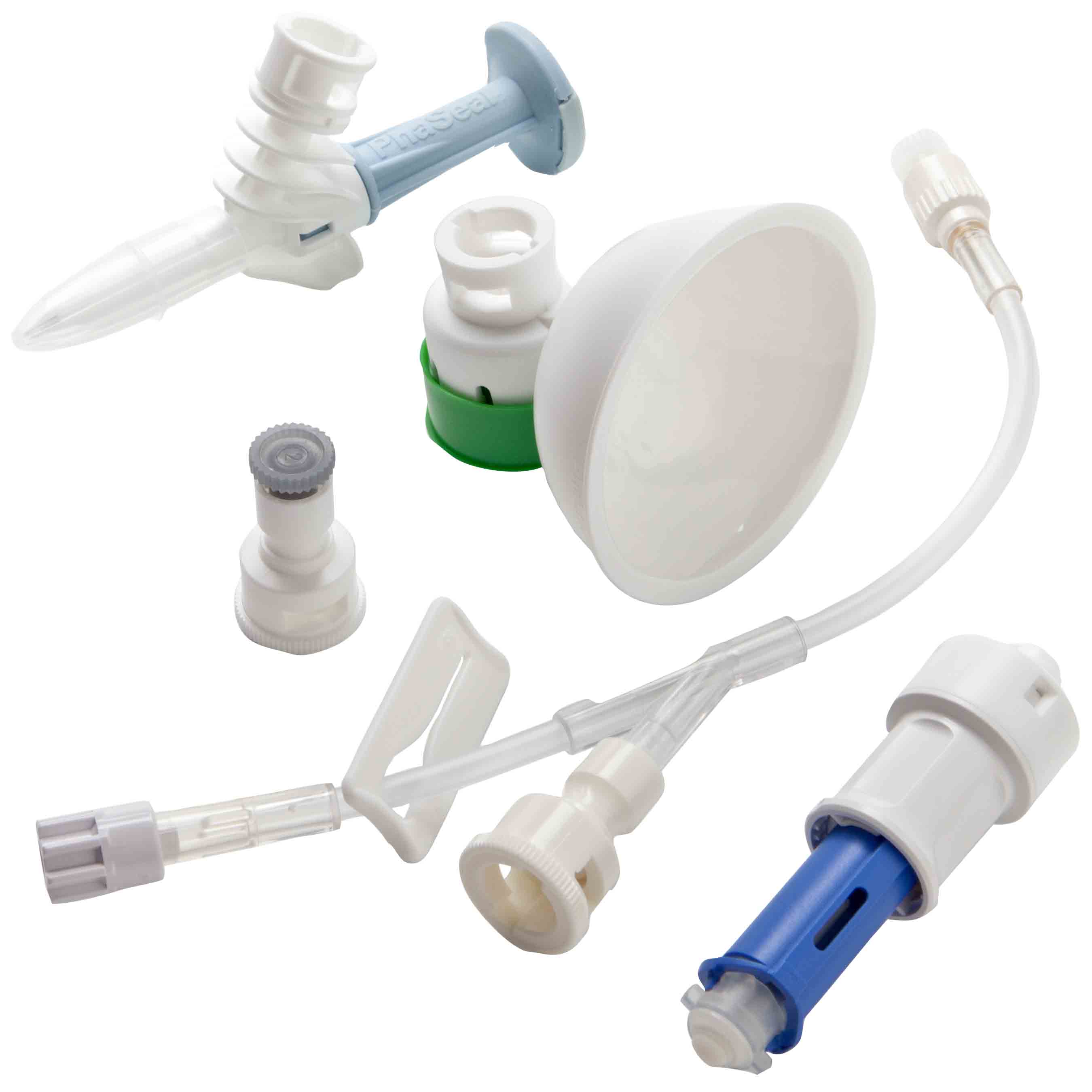
Important Update: BD® PhaSeal™ System and SmartSite™ VialShield Closed Vial Access Device Discontinuation
Why are the PhaSeal™ System and SmartSite™ VialShield devices being discontinued?
After careful consideration and in alignment with the BD commitment to innovation and safety, we have made the decision to retire the original PhaSeal™ Closed System Transfer Devices (CSTD) and the SmartSite™ VialShield Vial Access Devices. Based on current production schedules, we anticipate that inventory of the discontinued PhaSeal™ and SmartSite™ VialShield codes in the United States will be depleted by July 1, 2026.
Once inventory is depleted, we are confident that the BD® PhaSeal™ Optima System will continue to meet your hazardous drug preparation and administration safety needs.
Transitioning to PhaSeal™ Optima
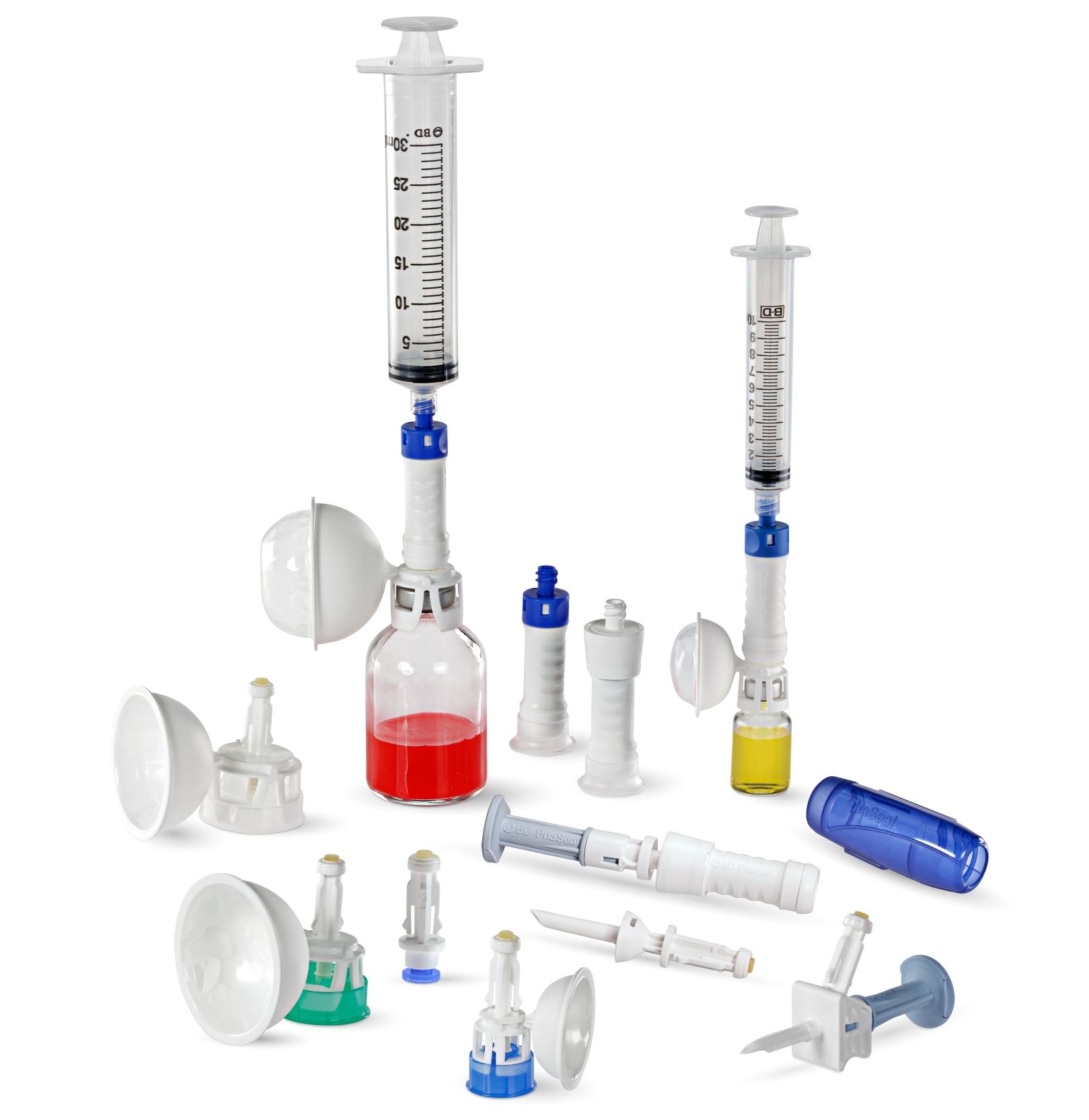
PhaSeal™ Optima: The Next Generation
BD has pioneered the CSTD category by listening to healthcare professionals who handle hazardous drugs every day. The PhaSeal™ Optima System builds on the proven foundation of the original PhaSeal™ System, maintaining its core architecture and physical barrier technology to prevent exposure. This includes a vapor-capturing mechanism in the vial protector and membrane-to-membrane connections that self-seal upon disconnect, ensuring airtight, leakproof transfers.
*Within an ISO Class V environment following aseptic technique
†The ability to prevent microbial ingress for up to 168 hours should not be interpreted as modifying, extending, or superseding a manufacturer’s labeling recommendations for the storage and expiration dating of the drug vial. Refer to the drug manufacturer’s recommendations and USP compounding guidelines for shelf life and sterility information.
Our collection of literature on industries and on our offerings gives you information you can use to continue striving for excellence.